![]() Click to view page images in PDF format.
Click to view page images in PDF format.
Composition and Origins of Coalbed Gas
Dudley D. Rice, U.S. Geological Survey
Search and Discovery Article #40009 (2000)
Adapted for online presentation from an article of the same title by the same author (p. 159-184) in AAPG Studies in Geology No. 38, Hydrocarbons from Coal, 1993, which is now available on CD-ROM in AAPG/Datapages Re-Discovery Series No. 1, along with Studies in Geology No. 17, Coalbed Methane Resources of the United States, and miscellaneous AAPG articles on the subject of coalbed methane (www.aapg.org/digital publications).
Coal, which contains more than 50% by weight and 70% by volume of carbonaceous material including inherent moisture (Bates and Jackson, 1987), has been and remains the most abundant energy source in the world. Reserves are estimated to be in the range of several trillion tons (Landis, this volume). Thus coal potentially represents a much larger energy resource than that estimated for crude oil and natural gas. In addition to minable reserves, coal has been and is being studied as a source of hydrocarbons, in particular natural gas. The presence of methane-rich gas in coal has long been recognized because of explosions that have occurred during underground mining. Not only is the gas a hazard, but coal mining also contributes to the increasing amount of atmospheric methane, which is a potent greenhouse gas (Boyer et al., 1990). In addition, coal is the source of hydrocarbons that have accumulated in adjacent reservoirs in many basins. Only recently has gas in coal beds been recognized as a large untapped energy resource with the coal serving as both the source and reservoir rock.
After burial, plant material is progressively converted to coal by the process of coalification (Levine, this volume). Figure 1 shows the rank sequence from peat to anthracite and common properties for measuring it. The alteration is controlled by biochemical processes during peat formation and by temperature and pressure during diagenesis and metamorphism. In addition to temperature and pressure, which are usually dependent on depth of burial, the conversion is determined by geologic time. Coalification changes physical and chemical properties, including increased calorific value and fixed carbon, decreased volatile matter, and moisture content, and also results in natural gas generation, the subject of this paper. In addition, coal beds, depending on their composition, are also source rocks for liquid hydrocarbons (condensate and oil) (Boreham and Powell, 1993; Clayton, 1993; Mukhopadhyay and Hatcher; 1993).
In the United States, there has been active exploration for coalbed gas because of a federal tax credit given for the production of gas from coal beds. In-place coalbed gas resources of about 400 trillion cubic feet (Tcf) have been estimated for the major U.S. coal-bearing basins shown on Figure 2 (ICF Resources, 1990). Of this resource, 90 Tcf are considered to be recoverable (ICF Resources, 1990). In comparison, the undiscovered recoverable resources of conventional gas in the United States are estimated to be 399 Tcf (U. S. Geological Survey and Minerals Management Service, 1989). The resulting exploration and production activity has resulted in new coalbed gas compositional data from desorption tests and production. These supplement compositional data from other parts of the world that were mainly collected during underground mining activities.
The objectives of this paper are to: (1) describe processes of natural gas generation, storage, and migration in coal beds, (2) distinguish the chemical and isotopic composition of coalbed gas, (3) relate composition to various factors, such as coal rank and composition, and secondary processes, such as mixing, oxidation, and migration and, (4) describe the occurrence, composition, and origin of coalbed gas in several areas around the world. An understanding of the compositional variations of coalbed gas and their controls is important for: (1) evaluating the economic value of coalbed gas, (2) identifying coal-derived gas that has migrated into adjacent reservoirs, and (3) resolving legal problems such as ownership of natural gas or liability for hazards associated with gas venting.
It is emphasized that this paper is on gases contained in or produced from coal beds, which are referred to as coalbed gases, and not gases from adjacent reservoirs that are interpreted to be coal-derived.
List of Figures Figure 1. Stages of coalification and common properties of measurement. Modified from
Stach et al. (1982). D.a.f.=dry and ash free.
Figure 1. Stages of coalification and common properties of measurement. Modified from
Stach et al. (1982). D.a.f.=dry and ash free.
![]() Figure 2. Map showing principal coal-bearing basins of the United States and estimates of
in-place coalbed gas resources. Coals in basins of central and eastern United States are of
Paleozoic age and those in western U.S. basins are of Cretaceous and Tertiary age.
Modified from ICF Resources (1990).
Figure 2. Map showing principal coal-bearing basins of the United States and estimates of
in-place coalbed gas resources. Coals in basins of central and eastern United States are of
Paleozoic age and those in western U.S. basins are of Cretaceous and Tertiary age.
Modified from ICF Resources (1990).
 Figure 3. Calculated amounts of gases generated from coal during coalification. Modified
from Hunt (1979).
Figure 3. Calculated amounts of gases generated from coal during coalification. Modified
from Hunt (1979).
 Figure 4. Van Krevelen diagram showing types of kerogen and pathways for generation of
methane and elimination of carbon dioxide and water during coalification. Liptinite
corresponds to types I and II kerogen, vitrinite to type III, and inertinite to type IV.
Modified from Levine (1987).
Figure 4. Van Krevelen diagram showing types of kerogen and pathways for generation of
methane and elimination of carbon dioxide and water during coalification. Liptinite
corresponds to types I and II kerogen, vitrinite to type III, and inertinite to type IV.
Modified from Levine (1987).
 Figure 5. Plot showing volumes of methane generated and stored per gram of coal with increasing
rank. Modified from Meissner (1984) and P.D. Jenden (personal communication, 1992).
Figure 5. Plot showing volumes of methane generated and stored per gram of coal with increasing
rank. Modified from Meissner (1984) and P.D. Jenden (personal communication, 1992).
 Figure 6. Methane d13C versus wetness (C2+) for coalbed gases in Canada and the United
States. Sources of data are listed in Table 1. Compositional fields are from Schoell (1983).
OM, organic matter.
Figure 6. Methane d13C versus wetness (C2+) for coalbed gases in Canada and the United
States. Sources of data are listed in Table 1. Compositional fields are from Schoell (1983).
OM, organic matter.
 Figure 7. Methane d13C versus methane
dD for coalbed gases in Poland and United States.
Sources of data are listed in Table 1. Compositional fields modified from Whiticar et al.
(1986). Composition of biogenic gases is also affected by d13C of carbon dioxide substrate
for those gases generated by carbon dioxide reduction and by dD of associated water for
those gases generated by methyl-type fermentation.
Figure 7. Methane d13C versus methane
dD for coalbed gases in Poland and United States.
Sources of data are listed in Table 1. Compositional fields modified from Whiticar et al.
(1986). Composition of biogenic gases is also affected by d13C of carbon dioxide substrate
for those gases generated by carbon dioxide reduction and by dD of associated water for
those gases generated by methyl-type fermentation.
 Figure 8. Methane d13C versus ethane
d13C for coalbed gases in Canada, Poland, and the
United States. Sources of data are listed in Table 1. Compositional trend for gases
generated from oil-prone source rocks with maturation is estimated from James (1983),
Sundberg and Bennett (1983), Jenden et al. (1988), and Jenden and Kaplan (1989).
Figure 8. Methane d13C versus ethane
d13C for coalbed gases in Canada, Poland, and the
United States. Sources of data are listed in Table 1. Compositional trend for gases
generated from oil-prone source rocks with maturation is estimated from James (1983),
Sundberg and Bennett (1983), Jenden et al. (1988), and Jenden and Kaplan (1989).
 Figure 9. Vitrinite reflectance (R0) versus gas wetness (C2+) for coalbed gases in Canada
and the United States. Sources of data are listed in Table 1.
Figure 9. Vitrinite reflectance (R0) versus gas wetness (C2+) for coalbed gases in Canada
and the United States. Sources of data are listed in Table 1.
 Figure 10. Vitrinite reflectance (R0) versus gas wetness (C2+) for coals and coalbed gases,
respectively, in eastern China. Line 1 is for coalbed gases from shallow coal mines; line 2 is
from medium-deep coal mines; line 3 is from deep boreholes; and line 4 is from pyrolysis.
From Dai et al. (1987).
Figure 10. Vitrinite reflectance (R0) versus gas wetness (C2+) for coals and coalbed gases,
respectively, in eastern China. Line 1 is for coalbed gases from shallow coal mines; line 2 is
from medium-deep coal mines; line 3 is from deep boreholes; and line 4 is from pyrolysis.
From Dai et al. (1987).
 Figure 11. Methane d13C versus vitrinite reflectance (R0) for coalbed gases and coals,
respectively. (A) Eastern China. Data from Dai et al. (1987). (B) Western Germany. Data
from Colombo et al. (1970). (C) Lower Silesian basin, Poland. Data from Kotarba (1988,
1990). (D) Canada and United States. Sources of data are listed in Table 1. Regression line
1 is for gases generated from types II/III kerogen (Faber, 1987); line 2 is for coalbed and
coal pyrolysis gases in China (Dai et al., 1987); line 3 is for coal-derived gases in China
(Shen et al., 1988); and stippled area is for coal-derived and coal pyrolysis gases from
several authors as summarized by Whiticar (1992).
Figure 11. Methane d13C versus vitrinite reflectance (R0) for coalbed gases and coals,
respectively. (A) Eastern China. Data from Dai et al. (1987). (B) Western Germany. Data
from Colombo et al. (1970). (C) Lower Silesian basin, Poland. Data from Kotarba (1988,
1990). (D) Canada and United States. Sources of data are listed in Table 1. Regression line
1 is for gases generated from types II/III kerogen (Faber, 1987); line 2 is for coalbed and
coal pyrolysis gases in China (Dai et al., 1987); line 3 is for coal-derived gases in China
(Shen et al., 1988); and stippled area is for coal-derived and coal pyrolysis gases from
several authors as summarized by Whiticar (1992).
 Figure 12. Depth versus methane d13C values for coalbed gases, southern Sydney basin,
Australia. Modified from Smith et al. (1985a).
Figure 12. Depth versus methane d13C values for coalbed gases, southern Sydney basin,
Australia. Modified from Smith et al. (1985a).
 Figure 13. Depth from top of Carboniferous versus methane d13C for coalbed gases from
western Germany. Data from Colombo et al. (1970).
Figure 13. Depth from top of Carboniferous versus methane d13C for coalbed gases from
western Germany. Data from Colombo et al. (1970).
 Figure 14. Variation in d13C values of methane desorbed from lump coal. From Smith et al.
(1985b).
Figure 14. Variation in d13C values of methane desorbed from lump coal. From Smith et al.
(1985b).
![]() Figure 15. Schematic diagram showing zones of alteration and original gas and their
characteristics and controls.
Figure 15. Schematic diagram showing zones of alteration and original gas and their
characteristics and controls.
 Figure 16. Carbon dioxide d13C versus carbon dioxide-methane index (CDMI) for coalbed
gases in Lower Silesian basin, Poland. From Kotarba (1990).
Figure 16. Carbon dioxide d13C versus carbon dioxide-methane index (CDMI) for coalbed
gases in Lower Silesian basin, Poland. From Kotarba (1990).
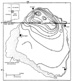 Figure 17. Map of San Juan basin, United States, showing isoreflectance (R0) lines on
Upper Cretaceous Fruitland Formation coal beds. From Law (1992).
Figure 17. Map of San Juan basin, United States, showing isoreflectance (R0) lines on
Upper Cretaceous Fruitland Formation coal beds. From Law (1992).
Click here to view animation of Figures 17, 18, and 19.
 Figure 18. Map of San Juan basin, United States, showing wetness (C2+) of Upper
Cretaceous Fruitland Formation coalbed gases. Modified from Scott et al. (1991).
Figure 18. Map of San Juan basin, United States, showing wetness (C2+) of Upper
Cretaceous Fruitland Formation coalbed gases. Modified from Scott et al. (1991).
Click here to view animation of Figures 17, 18, and 19.
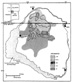 Figure 19. Map of San Juan basin, United States, showing carbon dioxide content of Upper
Cretaceous Fruitland Formation coalbed gases. Modified from Scott et al. (1991).
Figure 19. Map of San Juan basin, United States, showing carbon dioxide content of Upper
Cretaceous Fruitland Formation coalbed gases. Modified from Scott et al. (1991).
Click here to view animation of Figures 17, 18, and 19.
GENERATION OF COALBED HYDROCARBONS
STORAGE AND MIGRATION OF COALBED GAS
CHARACTERISTICS OF COALBED GASES FROM SELECTED AREAS
Bowen and Sydney Basins, Australia
San Juan Basin, New Mexico and Colorado, USA
Powder River Basin, Montana and Wyoming, USA
Black Warrior Basin, Alabama and Mississippi, USA
GENERATION OF COALBED HYDROCARBONS
Natural gas is generated in coal by two distinct processes: biogenic and thermogenic. Biogenic gas, primarily methane and carbon dioxide, is produced by the decomposition of organic matter by microorganisms and is commonly generated in peat swamps (Kim and Douglas, 1972). The breakdown of organic matter leading to methanogenesis is performed in a complex series of processes by a diverse population of microbes, each of which contributes to the partial oxidation of organic matter. Methanogenic microorganisms, which belong to the Archaea domain (Woese et al., 1990), only operate during the final stages of anaerobic decomposition of organic matter and depend on other bacteria to convert the complex organic compounds into simple precursors. The generation and accumulation of biogenic gas in ancient sequences is described by Rice and Claypool (1981), Zhang and Chen (1985), and Rice (1992).
The major requirements for the generation of significant amounts of biogenic gas are: anoxic environment, low sulfate concentrations, low temperatures, abundant organic matter, high pH values, adequate pore space, and rapid sedimentation (Rice and Claypool, 1981; Zhang and Chen, 1985; Rice, 1992). If these conditions are met, economically significant amounts of biogenic methane can be generated over a period of tens of thousands of years after burial (Claypool and Kaplan, 1974).
Two pathways have been identified for the generation of biogenic gas: carbon dioxide reduction and methyl-type fermentation (Schoell, 1980; Woltemate et al., 1984; Jenden and Kaplan, 1986; Whiticar et al., 1986). Most species have the ability to generate methane by carbon dioxide reduction; carbon dioxide is available from both early bacterial processes and thermal decarboxylation of organic matter (Carothers and Kharaka, 1980). Fermentation is performed by a few species of bacteria by the decarboxylation of several substrates, but commonly acetate, whereby the methyl group is converted to methane and the carboxyl group is converted to carbon dioxide.
Although both pathways are operating in near-surface, recent environments, most ancient accumulations have probably resulted from carbon dioxide reduction based on chemical and isotopic composition (Rice, 1992). Jenden and Kaplan (1986), Coleman et al. (1988), and Schoell (1988) suggested that depth of burial and the age of organic-rich sediments regulate the relative significance of these two pathways. Fresh, near-surface sediments can generate biogenic gas by both pathways, but most of this early-generated gas is probably lost to the atmosphere and is not a major contributor of ancient accumulations. In contrast, altered, more deeply buried sediments can generate biogenic gas mainly by carbon dioxide reduction and this gas would have a better chance of being preserved.
Biogenic gas can be generated during two different stages in coal beds. Biogenic gas is formed early in the burial history of low-rank coal (peat to subbituminous rank-R0 values <0.5%), and its generation and preservation are favored by rapid deposition; this biogenic gas is referred to as early stage. Most accumulations of ancient biogenic gas were probably generated during this early stage (Rice and Claypool, 1981; Rice, 1992). In addition, biogenic gas in coal beds can also be generated in recent geologic time (tens of thousands to a few million years ago) and is associated with active groundwater systems; this type of biogenic gas is referred to as late stage. The generation and occurrence of biogenic gas in groundwater systems has been documented by Carothers and Kh raka (1980), Barker and Fritz (1981), Rice and Threlkeld (1982), Coleman et al. (1988), and Grossman et al. (1989). Coal beds are commonly regional aquifers (Kaiser et al., 1991; Tyler et al., 1991), which provide favorable environments for bacterial activity, including methane generation. This late-stage biogenic gas generation can take place in coal beds of any rank provided that the requirements outlined previously have been met. The initial microbial process in the aquifer is probably aerobic oxidation, which gives rise to a new food supply for the anaerobic community that can start functioning when the dissolved oxygen in the water is eliminated. Late-stage biogenic methane generation is due mainly to reduction of carbon dioxide produced in previous oxidation phase by electrons (hydrogen) generated by fermentation of the other oxidized substrates produced by earlier oxidation.
With increasing coalification resulting from higher temperatures and pressures, coals become enriched in carbon as large amounts of volatile matter rich in hydrogen and oxygen are released. Methane, carbon dioxide, and water are the most important byproducts of this devolatilization process (Stach et al., 1982) (Figure 3). The generation of methane and associated hydrocarbons is thermal in origin and occurs at ranks of high-volatile bituminous and higher (R0 values > 0.6%). The quantities of these main volatile substances (methane, carbon dioxide, and water) can be estimated using the major elemental composition (C-H-O) (Juntgen and Karweil, 1966; Karweil, 1969; Juntgen and Klein, 1975; Levine, 1987). A van Krevelen diagram (Figure 4) shows the trends for the elimination of carbon dioxide and water, and for methane generation from different types of kerogen during progressive stages of coalification.
The four types of kerogen shown in Figure 4 are determined by elemental analysis (Tissot and Welte, 1984) and generally correlate with the major maceral groups in coal identified by petrography. Liptinite-rich coals generally correspond to types I and II kerogen, vitrinite-rich coals to type III kerogen, and inertinite-rich coals to type IV kerogen.
Significant amounts of methane are generated from coals during the entire coalification process. The estimates are variable (about 100 to 300 cm3/g of coal) depending on the elemental data employed, starting rank, and assumptions made about the products (Juntgen and Karweil, 1966; Juntgen and Klein, 1975; Hunt, 1979; Meissner, 1984; Welte et al., 1984; Levine, 1987). In addition, the methane yield is influenced by the coal composition (Juntgen and Karweil, 1966; Levine, 1987). The actual yield in nature is probably in the range of 150 to 200 cm3/g of coal.
The liptinite maceral is generally hydrogen-rich and is known to be capable of generating oil (Boreham and Powell, 1993; Mukhopadhyay and Hatcher, 1993). However, most coals are classified as humic and are composed of more than 80 to 90% of the maceral vitrinite, which is considered to be composed of oxygen-rich type III kerogen. Recent petrographic and geochemical studies have indicated that certain components of the vitrinite maceral group are hydrogen-rich and are also capable of generating liquid hydrocarbons (Bertrand et al., 1986; Saxby et al., 1986; Saxby and Shibaoka, 1986; Khorasani, 1987; Rice et al., 1989a, 1992; Lu and Kaplan, 1990; Clayton et al., 1991). Because liptinite and hydrogen-rich vitrinite have been shown to be source rocks for oil, they are also considered to be the source rocks for wet gas; that is, gas that contains significant amounts of heavier hydrocarbons.
Major quantities of carbon dioxide are generated by thermal decarboxylation or devolatilization of coal particularly prior to the main stage of thermogenic methane production (Karweil, 1969; Hunt, 1979; Tissot and Welte, 1984) (Figure 3). However, carbon dioxide is highly soluble in water and very reactive and has different origins (Hunt, 1979). As a consequence, the carbon dioxide presently in a coalbed gas may have an origin other than devolatilization. Other possible sources of carbon dioxide in coalbed gas are (1) thermal destruction of carbonates (Hunt, 1979), (2) carbonate dissolution linked to silicate hydrolysis (Smith and Ehrenberg, 1989), (3) bacterial degradation of organic matter (Carothers and Kharaka, 1980; Whiticar et al., 1986), (4) bacterial hydrocarbon oxidation (Jam s and Burns, 1984), and (5) migration from magma chambers or crust (Smith et al., 1985a; Kotarba, 1988, 1990).
STORAGE AND MIGRATION OF COALBED GAS
Coalbed gas is stored in the coal (1) upon (adsorbed) or within (absorbed) the molecular structure of the coal or (2) within the micropores and cleats (Juntgen and Karweil, 1966). Because coal has large internal surface areas (van Krevelen, 1981), it is thought that the gas is generally retained by molecular attraction on these surfaces. However, the microstructure of the coal has been described as a molecular sieve or cage (van Krevelen, 1981) within which the methane molecule can be stored.
As discussed previously, the ability of coal to generate thermogenic gas is mainly the function of rank, and the rates of generation rapidly increase with rank. The ability of coal to store gas is also a function of rank, and the storage capacity drops off considerably with increasing rank (Juntgen and Karweil, 1966; Meissner, 1984) (Figure 5). In addition to rank, total methane storage is affected by pressure and temperature; increasing pressure results in increased storage capacity, whereas increased temperature results in decreased storage capacity (Meissner 1984; Wyman, 1984). At higher ranks (medium-volatile bituminous rank and higher), coals may have generated more methane than they can store (Figure 5). This is an important point to consider when evaluating the gas content and possible expulsion of the coalbed gas into adjacent reservoirs.
Levine (1991) made the case that the gas storage capacity of coal with hydrogen-rich kerogen (either liptinite or hydrogen-rich vitrinite) is adversely affected by the presence of higher molecular weight volatile constituents, such as oil. The molecular structure of the coal can generally accommodate small molecules such as methane, carbon dioxide, and water. However, the micropore structure may be "plugged" by liquid hydrocarbons in high-volatile bituminous coals, thus reducing the storage capacity (Thomas and Damberger, 1976). Removal of these plugs and increased sorption capacity results from the thermal cracking of these volatiles at higher ranks (low-volatile bituminous to anthracite) (Thomas and Damberger, 1976).
Because of the strong adsorption affinity for C2+ hydrocarbons relative to methane, liquid hydrocarbons do not easily migrate out of coals (Wyman, 1984; Saxby and Shibaoka, 1986; Hunt, 1991; Littke and Leythaeuser, this volume). These heavier hydrocarbons are probably thermally cracked to lighter hydrocarbons, including natural gas, and this cracking process has a significant impact on the composition, both molecular and isotopic, of the natural gas.
Natural gas is held in the microstructure of coal by reservoir pressure. Gas migration by desorption, diffusion, and free-phase flow takes place when the pressures in the coal beds are decreased, which can take place either naturally or by human activities. The natural method of depressurization is by uplift and erosion, which commonly happens along the margins of basins, but can also occur over larger areas. Pressure reduction in coal beds also results from either coal mining or from gas production from wells. Because most coals are characterized by high water saturations, depressurization results from dewatering of the coal beds. The release and subsequent migration of the gas by desorption and diffusion processes through the microstructure of the coal can result in alteration of the coalbed gas composition as will be discussed later.
The principles of hydrocarbon generation, storage, and migration in coal, as illustrated in Figure 5, are important for three reasons. First, the potential in-place resource of coalbed gas can be estimated. Second, a prediction of the amount of coal-derived gas that might have migrated out of the coal beds can be made. This migrated gas has either accumulated into adjacent reservoirs and is an important resource, or has been lost to the atmosphere. Finally, migration of gas within the coal bed can significantly alter the composition of the gas.
Until recently, the source of coalbed gas compositional data was mostly from underground mines. With the recent exploration activity for coalbed gas in the United States driven by a production tax credit, gas composition analyses are now available from two additional sources away from mining areas: desorption tests of coal samples and production from coalbed reservoirs. Gas desorption tests are conducted for the primary purpose of evaluating the gas content of the coal. Samples, generally core and sometimes cuttings, are quickly placed in an air-tight canister and desorbed over a period of several days to months (Close and Erwin, 1989). The released gas is usually measured for volume and sometimes analyzed for composition.
Available information on the composition of natural gas from coal beds is given in Table 1 and displayed in Figures 6 through 11. Information is generally provided only where both molecular and isotopic data are available. Data are for gas samples collected from coal beds and are not for gas samples from adjacent reservoirs that are interpreted to have been coal-derived (for example, Schoell, 1980; Liu et al., 1987; Shen et al., 1988). The analyzed gases were collected from coals ranging in age from Pennsylvanian (Late Carboniferous) to Tertiary and in rank from lignite to anthracite (R0 values of 0.3 to 4.9%). The depth of the gaseous coals extends from about 400 ft (underground mines and shallow gas wells) to 14,500 ft (boreholes). Coalbed gases from the United States are mainly production gases, whereas those from Australia, China, Germany, and Poland are generally associated with underground coal mines, although a few samples were obtained from deeper boreholes.
The molecular and isotopic composition of coalbed gases from all localities is highly variable (Table 1). Methane is usually the major component with other hydrocarbon gases and carbon dioxide occurring in lesser amounts. The hydrocarbon composition of the gases, which is expressed as gas wetness (C2+) or the percentage of ethane and higher hydrocarbon gases, ranges from 0 to 70.5% (Table 1). The wettest gases occur in western Germany where the coal samples were both desorbed and crushed, and the composition is undoubtedly affected by the contribution of wet residual gas from the crushed rock. Carbon dioxide is the other major component of coalbed gases and varies from zero to more than 99%. Coalbed gases are also variable in their isotopic composition; methane d13C values range from about -80 to -16.8 o/oo; ethane d13C values range from -32.9 to -22.8 o/oo, methane dD values from -333 to -117 o/oo, and carbon dioxide d13C values from -26.6 to +18.6 o/oo (Table 1). The methane d13C values of desorbed gases are isotopically heavier by several permil than production or "free" gases (Colombo et al., 1970; Kotarba, 1988). This difference is the result of the desorption process which will be discussed later in the paper.
Figure 6 shows the relation between methane d13C values and gas wetness (C2+) for coalbed gas samples from Canada and the United States; this relation is considered to be typical for coalbed gases in other localities. Although many gas samples in Canada and the United States are chemically dry over the entire range of methane d13C values from about -61 to -29o/oo, the gas samples with intermediate methane d13C values in the range of -48 to -34o/oo are generally the wettest (Figure 6). In addition, gases from Paleozoic coals in central and eastern United States (Arkoma and Black Warrior basins) (Figure 2) are methane-rich at all ranks, whereas gases from Cretaceous coals of western basins (Piceance and San Juan basins) (Figure 2) are sometimes wet at intermediate methane d13C values.
The compositional fields for different types of gases as shown in Schoell (1983), including coal-derived gases, are plotted on the Figure 6. It is interesting to note that the coalbed gases from Canada and the United States plot within the field of gases generated from sapropelic organic matter. None of the samples shown in Figure 6 fall within the field for gases generated from humic organic matter, although some samples from China, Germany, and Poland do. Some of the wettest gases from the Piceance and San Juan basins are also associated with waxy oil (Clayton, this volume).
Methane dD and ethane d13C values are available for fewer coalbed gas samples than are methane d13C values. Methane becomes enriched in deuterium (more positive dD values) and the heavy isotope 13C (more positive d13C values) with increasing maturation (Figure 7). The methane and ethane d13C values for some of the coalbed gas samples become isotopically heavier with increasing maturation as predicted (Figure 8). However, many gas samples display a wide range of methane d13C values over a narrow range of ethane d13C values suggesting a complex origin for the gases.
The relation between gas wetness (C2+) and rank of associated coal (R0) is illustrated by coalbed gas samples from Canada, China, and the United States (Figures 9 and 10). The trends displayed by these figures are similar to that between methane d13C and gas wetness illustrated in Figure 6. Two observations can be made from the plots. First, there is a trend for all of the gases being methane-rich at low and high ranks and for at least some samples being wet at intermediate ranks. The wettest gases in Canada and the United States occur at R0 range of about 0.6 to 0.8%, whereas the wettest gases in China occur at R0 values of about 1.25 to 1.35%. Second, gases from Paleozoic coals (Arkoma and Black Warrior basins) are methane-rich at all ranks, whereas gases from Cretaceous coals (Piceance and San Juan basins) are wet at intermediate ranks. Finally, gases from Carboniferous, Permian, and Triassic through Tertiary coals in China from shallow underground mines are methane-rich at all ranks, whereas those from deeper horizons and from pyrolysis experiments are wetter in composition at intermediate ranks.
Figure 11 shows the relation between methane d13C values of coalbed gas and R0 of associated coal from Canada, China, Germany, Poland, and the United States. In general, methane d13C values are isotopically lighter at lower ranks and isotopically heavier at higher ranks. However, gases from coal of a given rank display a wide range of methane d13C values. In addition, the methane d13C values commonly fall above (isotopically lighter) the regression lines shown in Figure 11 which were determined for gases interpreted to have been generated from coal and different types of dispersed organic matter.
Although methane d13C and wetness (C2+) values of coalbed gases are scattered when plotted against the rank (R0) of the associated coal, they illustrate a more systematic trend when plotted against present-day depth of burial as illustrated by data from Australia, China, and Germany. There appears to be a widespread pattern in which shallow coalbed gases are composed of isotopically light methane, as compared with deeper coalbed gases, regardless of rank (Figures 10, 12, and 13). Also, the lightest methane d13C values for coalbed gases of a given rank for all countries shown in Figure 11 generally occur at the shallowest depths. The methane-rich nature of shallow coalbed gases has been known for years because the first source of compositional data was from underground coal mines, thus the term coalbed methane came into being. This change in molecular and isotopic composition with depth can take place over a transition zone, can be abrupt, and can take place at varying depths, but usually in the range of a few thousand feet.
Based on the molecular and isotopic composition of the coalbed gases and the rank of the associated coal (R0), the coalbed gases from around the world are both biogenic and thermogenic in origin. The primary controls of the molecular and isotopic composition of coalbed gases appear to be rank, composition, and depth/temperature of associated coal. Direct measurements or extrapolated values of rank for associated coals are commonly available. However, coal composition data, including both maceral and geochemical analyses, are less common, making the interpretation of the gas more difficult. Secondary controls are also reflected in the coalbed gas composition and will be discussed later.
Biogenic gas generated from all types of coal consists mainly of methane (Rice and Claypool, 1981; Schoell, 1983; Zhang and Chen, 1985; Rice, 1992). The presence of significant amounts of heavier hydrocarbon gases with biogenic methane indicates the overprint of late-stage biogenic methane in mature coals that have already generated thermogenic hydrocarbons. Biogenic gas generation is generally restricted to shallow depths and low temperatures (Rice and Claypool, 1981; Zhang and Chen, 1985; Rice, 1992) and can occur in coals ranging in rank from peat to anthracite.
Because methane-rich gases may have origins other than microbiological processes and biogenic gas can occur in coals over a wide range of ranks, methane d13C values provide evidence for its biogenic origin. In general, biogenic methane is isotopically light because of enrichment of the isotope 12C by microorganisms (methane d13C values generally in the range of -55 to -90o/oo) (Rice and Claypool, 1981; Rice, 1992). Biogenic gases can be further differentiated on the basis of their generation by the methyl-type fermentation and carbon dioxide reduction pathways (Jenden and Kaplan, 1986; Whiticar et al., 1986). However, no absolute ranges can be given for the d13C and dD values of methane resulting from these two pathways. Biogenic methane resulting from carbon dioxide reduction is generally isotopically light in carbon (d13C values in the range of -55 to -110o/oo), whereas methane derived from methyl-type fermentation can be isotopically heavy (d13C values in the range of -40 to -70o/oo) (Figure 7). However, the d13C values of methane produced by carbon dioxide reduction are strongly correlated to the d13C of the carbon dioxide substrate; the isotopic fractionation between the two species remains relatively constant at about 70 ± 10o/oo (Claypool and Kaplan, 1974; Jenden and Kaplan, 1986). As a consequence, biogenic methane as heavy as -40 o/oo can be produced by the reduction of isotopically heavy carbon dioxide (see for example, Jenden and Kaplan, 1986). Biogenic methane produced from carbon dioxide reduction is generally enriched in deuterium (dD values in range of -150 to -250o/oo) and methane derived from methyl-type fermentation is depleted in deuterium D values in range of -250 to -400o/oo). However, the source of the hydrogen in biogenic methane is the associated water, which is variable in isotopic composition (Jenden and Kaplan, 1986; Whiticar et al., 1986). As a result, the methane dD values are dependent upon formation water dD as well as the biochemical pathway. Based on data displayed in Table 1 and on Figure 7, some of the coalbed gases are biogenic and appear to have resulted from generation via both pathways, carbon dioxide reduction and methyl-type fermentation. However, this interpretation does not take into consideration the dD values of the associated water and d13C values of carbon dioxide substrate.
The co-existence of isotopically heavy ethane (ethane d13C values in the range of -28 to -24o/oo) with isotopically light methane (methane d13C values more negative than -55o/oo), as shown by Figure 8, indicates the mixing of thermogenic ethane and biogenic methane. This type of mixing is probably a good signature for identifying late-stage biogenic gas.
In contrast to biogenic gases, thermogenic coalbed gases are characterized by (1) common presence of heavier hydrocarbons (C2+ values can be several percent or greater) at intermediate ranks of high-volatile and medium-volatile bituminous coal, (2) enrichment of heavy isotope 13C in methane and ethane with increasing rank (methane d13C values more positive than -55o/oo and ethane d13C values more positive than -33o/oo), and (3) enrichment of deuterium (methane dD values more positive than -250o/oo) with increasing rank (Figures 6 through 8). Based on data by Michael et al. (in press), the onset of thermogenic hydrocarbon generation in coal beds starts at Ro val es of about 0.6%. However, economic quantities of thermogenic hydrocarbons may not be generated until R0 values of 0.7% (Tang et al., 1991). As shown by data from Juntgen and Karweil (1966), generation begins at lower levels for sapropelic coals than for humic coals.
At intermediate ranks (high-volatile to medium-volatile bituminous coal), sapropelic coals (types I and II kerogen-liptinite and hydrogen-rich vitrinite) contain both wet gas and liquids, whereas humic coals (type III kerogen-mostly vitrinite) contain a drier gas. At high ranks, coalbed gases are mostly methane, which results from generation of methane from residual kerogen and cracking of previously formed heavier hydrocarbons.
Thermogenic methane is isotopically more positive than biogenic methane because of the smaller kinetic isotope effects associated with thermal cracking. The principles governing the kinetic isotopic fractionation of thermogenic gases, including coalbed gases, are described by Chung et al. (1988), Clayton (1991), and Berner et al. (1992). In general, d13C values for thermogenic gases become isotopically heavier with increasing rank because residual gas-producing carbon becomes enriched in 13C due to 12C-12C bonds being broken more frequently by thermal processes than 12C-13C bonds. However, several authors, including James (1983, 1990), Rice (1983), Rice et al. (1989a), Johnson and Rice (1990), and Clayton (1991) have indicated the control of source rock composition, in addition to thermal maturity, on the isotopic composition of natural gases. In general, gases generated from coals with oxygen-rich kerogen (mostly vitrinite) (1) have more positive methane d13C values than those generated from coals with more hydrogen-rich kerogen (liptinite and some hydrogen-rich vitrinite) at similar levels of thermal maturity, and (2) exhibit less spread in methane and ethane d13C values. These differences result from isotopically lighter methane being produced by thermal cracking of aliphatic-type structures, which are prevalent in hydrogen-rich kerogen, whereas isotopically heavier methane results from the cracking of aromatic structures that are predominant in oxygen-rich kerogen (Galimov, 1988). This effect on isotopic composition is illustrated by regression lines for different types of kerogen shown on Figure 11.
Liquid hydrocarbons are generated at early stages of thermal cracking in coal (Lu and Kaplan, 1990). In noncoal lithologies with dispersed kerogen, such as shales, these liquid hydrocarbons are commonly expelled from the source beds and are not a factor in determining the composition of the gaseous hydrocarbons. In coals, however, these liquid hydrocarbons are commonly retained within the microstructure of the coal and are probably thermally cracked at higher temperatures to form gas. This thermal cracking process has an effect on the isotopic composition of the gases which is probably isotopically more negative than that for gases generated directly from kerogen (Smith et al., 1985b; Galimov, 1988).
As discussed previously, coal composition is probably a primary control on both the molecular and isotopic composition of coalbed gases. Phillips and Cross (1991) have documented changes in vegetation in peat swamps during geologic time with the most prominent change taking place in the Pennsylvanian (Late Carboniferous). Hunt (1991) noted that, although most of the world's ultimate reserves of coal are of Late Carboniferous and Permian age, most of the high wax, coal-derived oils are from reservoirs of Cretaceous and Tertiary age (see Clayton, this volume). These observations suggest that younger coals are probably more capable of generating wet gas than those of Paleozoic age. This conclusion is supported by differences in molecular composition of gases from the western United State versus central and eastern U.S. basins shown in Figure 6.
Secondary processes also affect the composition of coalbed gases, particularly at shallow depths. As discussed earlier, the main changes resulting from these secondary processes are that shallow coalbed gases, regardless of the rank of the associated coal, are composed mainly of isotopically light methane. The depth interval where these secondary processes are active and affect the composition of the gases is termed the zone of alteration and has been referred to in studies by Colombo et al. (1970) and Dai et al. (1987). Unaltered gas, generally in the deeper parts of basins, is referred to as original gas.
At shallow depths, coal beds are commonly aquifers where microorganisms can thrive. Microbial activity can affect the composition of coalbed gases in three ways. First, significant amounts of late-stage anaerobic biogenic gas can be generated that either can be mixed with previously generated thermogenic gas or can fill degassed coal beds. Biogenic gas is composed of isotopically light methane (Rice and Claypool, 1981; Schoell, 1983; Rice, 1992) and its mixing with earlier generated thermogenic gas could account for the compositional changes observed in shallow coal beds. Second, aerobic bacterial activity can result in the oxidation and consumption of methane. This process results in a kinetic isotope shift in the residual methane whereby both the carbon (d13C) and hydrogen (dD) isotopic ratios become more positive (Coleman et al., 1981; Whiticar and Faber, 1986; Alperin et al., 1988). Because methane is being consumed and because of the direction of shift of the isotope ratios, this process is not considered to be important in controlling the compositional changes observed in shallow coalbed gases. Finally, aerobic bacteria are capable of preferentially attacking the wet gas components (C2+) resulting in the destruction of most of the wet gases (James and Burns, 1984). As a result of the alteration, the d13C values of the residual propane and other wet gases are much heavier than expected. Bacterial alteration of the wet gas components could explain the molecular compositional changes observed in shallow coalbed gases.
Migration resulting from uplift, erosion, and depressurization is another possible secondary control on the composition of coalbed gas at shallow depths. Using experimental results and a mathematical simulation, Fuex (1980) concluded that there was negligible (<1o/oo) fractionation of carbon and hydrogen isotopic composition associated with migration of commercial gas accumulations. Fuex (1980) did not, however, evaluate the possible effects of isotopic fractionation related to desorption and diffusion of coalbed gas. Studies by Wyman (1984), Smith et al. (1985b), and Gould et al. (1987) have documented the significant "chromatographic" effect of desorption and diffusion on both the molecular and isotopic composition of coalbed gas. Desorbed gases become enriched in heavier hydrocarbons and carbon dioxide (if both methane and carbon dioxide are present), and methane and carbon dioxide d13C become isotopically heavier (13C-enriched) with increasing time. The magnitude of the compositional changes caused by desorption and diffusion is affected by the size of the system and the relative extent of desorption. For example, Hale and Firth (1988) showed that the molecular composition of a coalbed gas produced from a well over a period of 27 years remained essentially unchanged, whereas variation of methane d13C values during desorption of a lump of coal is significant (Figure 14). Finally, the changes in both isotopic and molecular composition resulting from desorption and diffusion are not consistent with the changes generally observed in shallow coalbed gases. Therefore, migration is not considered to be an important secondary process. Nevertheless, migration does result in degassing of the original coalbed gas, which is commonly thermogenic, and the remaining gas is commonly mixed with late-stage biogenic gas.
The zone of alteration, in which mixing and oxidation affect the coalbed gas composition, can extend from depths of a few hundred to a few thousand feet. The zone is usually restricted to the margins of basins; however, it can extend into and throughout a basin under certain conditions. The primary controls of the depth and lateral extent of the zone of alteration are the physical nature of the coal beds, burial history, and hydrology. Thick, laterally continuous, permeable coal beds are conducive to the development of an aquifer system. Uplift and erosion result in depressurization of the coal beds and subsequent degassing of the original gas. In addition, uplift and erosion result in relaxation of the cleat system and increased permeability, thus aiding both aquifer development and degassing. Finally, microorganisms become widely distributed in active aquifer systems leading to widespread biological activity, including aerobic oxidation and anaerobic methanogenesis. The concept of the zone of alteration versus original gas is diagrammatically illustrated in Figure 15.
In addition to hydrocarbon gases, carbon dioxide is the other significant component of coalbed gases. Carbon dioxide content varies from 0 to greater than 99%, and carbon dioxide d13C values, where analyses are available in Australia, Poland, and the United States, range from -26.6 to +18.6o/oo (Table 1; Figure 16). The wide range in concentrations and isotopic composition (d13C) suggests that significant amounts of carbon dioxide in coalbed gas are commonly not a product of the coalification process, but rather of other processes active in localized areas. The concentration and isotopic composition (d13C) of carbon dioxide in coalbed gases vary within a basin and from basin to basin indicating different origins (Figure 16). In addition, the concentration and isotopic composition (d13C) of carbon dioxide in coalbed gases is commonly different from those in gases of adjacent reservoirs suggesting different origins. Because of complexity of its occurrence and origin, the carbon dioxide in coalbed gases can best be interpreted by studying individual areas.
CHARACTERISTICS OF COALBED GASES FROM SELECTED AREAS
General interpretations of the composition and origins of coalbed gases have been made based mainly on the molecular and isotopic composition of the gases and rank of the associated coals. Because of the influence both of primary controls (coal rank and composition and depth/temperature) and secondary processes (mixing and oxidation) the coalbed gases can be better understood if individual areas are studied.
Colombo et al. (1970) conducted one of the first major studies characterizing the molecular and carbon isotopic composition of coalbed gases with their samples coming from the Aachen, Lower Rhine, and Saar coal-mining districts of western Germany. Also analyzed were coalbed gases from an adjacent deep borehole. Both "free" and "adsorbed" gases were collected and analyzed. "Free" gases were those collected prior to and during crushing of coal samples in an airtight container. "Adsorbed" gases were collected later from the same coal samples subjected to vacuum and heat in an air-tight container. In general, "free" methane was about 5o/oo more negative than "adsorbed" methane.
The coals in western Germany are of Late Carboniferous age (Westphalian and Stephanian) and range in rank from high-volatile A bituminous to anthracite (R0 values of 0.8 to 4.9%). They are composed mostly of vitrinite (69 to 90%) with lesser amounts of inertinite (3 to 28%) and liptinite (5 to 26%).
The coalbed gases vary considerably in both molecular and isotopic composition, which appears to be typical for mining areas (Table 1) (Colombo et al., 1970). The wetness (C2+) of the gases varies from 0 to 70.5% and methane d13C values range from -70.4 to -16.8o/oo; the methane d13C value of -16.8o/oo is the heaviest reported for a coalbed gas. Figure 11B illustrates that there is considerable scatter in methane d13C values versus rank (R0), and many methane d13C values are isotopically lighter at a given R0 value than those indicated by the regression lines. Coals from the Aachen and Lower Rhine districts are generally higher in rank, but commonly contain isotopically lighter methane than those from the Saar district; this relation would not be expected if rank were the main control of isotopic composition. Figure 13 shows a plot of methane d13C values versus depth relative to top of the Carboniferous datum for the coalbed gases. As demonstrated by Figure 13, methane d13C values generally become isotopically heavier with depth, regardless of rank. However, in the Saar district, the methane in the Ensdorf mine is isotopically lighter than that from the Gottelborn mine, in spite of the fact that the associated coals from the two mines are about the same rank and occur at about the same depth.
Colombo et al. (1970) considered the strong relation between coalbed gas composition (methane d13C and wetness) and depth to be the result of migration. However, the author interprets the changes in coalbed gas composition to be the result of secondary processes, such as mixing of biogenic gas and oxidation of wet gases, in the zone of alteration. In western Germany, the main coalification process and thermogenic hydrocarbon generation took place in Carboniferous time. In Early Permian time, folding, uplift, and erosion resulted in degassing of the coal beds. Relatively recently, the coalbed gases have probably been altered in conjunction with groundwater flow. The zone of alteration is about 600 ft deeper in the Aachen and Lower Rhine districts, as compared to the Saar district, which probably explains the isotopically lighter, methane-rich gases in those districts. The difference in the gas composition between coalbed gases in the Ensdorf and Gottelborn mines of the Saar district, where the coals are about the same rank and depth, can be explained by the presence of different sealing conditions as described by Colombo et al. (1970). At Ensdorf, the coal beds are sealed by Triassic claystones, whereas at Gottelborn this seal is missing and groundwater can easily enter the coal.
A detailed study of different mines showed that the zone of alteration is not horizontal (Colombo et al., 1970). The depth of the zone was determined to occur at different depths and to be controlled by permeability, seals, tectonics, and attitude of the beds.
The coalbed gases from the deep borehole and a couple of mine locations are characterized by isotopically very heavy methane d13C values (as much as -16.8o/oo). This isotopically heavy methane could be the result of two factors. First, degassing of coals by desorption and diffusion in late Paleozoic time might have resulted in the preferential loss of the isotopically lighter methane from the deeper horizons. As a result, the residual gas would be isotopically heavier in methane than the original gas. Second, regeneration of thermogenic methane may have taken place when the Carboniferous coals were later covered by Triassic sediments after Permian uplift and erosion. This second-stage thermogenic methane would be isotopically heavier because it was generated from residual carbon that was enriched in 13C because of earlier thermogenic gas generation.
Bowen and Sydney Basins, Australia
Extensive studies on the molecular and isotopic composition of coalbed gases in the mining areas of Bowen and Sydney basins of Australia have been carried out for the purposes of determining the distribution and origins of the gas as an aid in predicting outbursts and in utilization. Summaries of the studies are given by Smith et al. (1985a, b) and Gould et al. (1987). The gas samples were generally desorbed from coal cores. The effects of desorption and diffusion on the molecular and isotopic composition of coalbed gases, as described earlier in this paper, are given in Smith et al. (1985b) and Gould et al. (1987).
The coals of the Bowen and Sydney basins are of Permian age and range in rank from high-volatile B bituminous to medium-volatile bituminous (R0 values of 0.7 to 1.2%). The associated coalbed gases vary considerably in both molecular and isotopic composition (Table 1) (Smith et al., 1985a, b). Wetness (C2+) values of the gases are as much as 10% and carbon dioxide contents are as much as 99%. Methane d13C values range from about -80 to -24o/oo, methane dD values range from -279 to -171o/oo, and carbon dioxide d13C values range from -24.9 to 16.7o/oo.
High carbon dioxide contents characterize the coalbed gases from both basins. Although the carbon dioxide displays a wide isotopic range, the highest concentrations have a d13C value of about -7o/oo, which indicates a magmatic origin (Smith et al., 1985a). The lower amounts in each basin have a distinctly different isotopic composition indicating another origin. In the Collinsville mining area of the Bowen basin, carbon dioxide content increases with depth. In contrast, carbon dioxide contents in the southern Sydney basin vary both laterally and between coal beds penetrated by a single borehole (Smith et al., 1985a). The highest concentrations of carbon dioxide are localized laterally along major faults. Smith et al. (1985a) considered the interbed variations to be a function of faults and varying permeabilities between coal beds that may be controlled by maceral composition. The carbon dioxide first moved vertically along faults and then migrated along coal beds with higher permeability.
In the Collinsville mining area (Bowen basin), trends in hydrocarbon gas composition, in addition to carbon dioxide, are related to depth of the coal beds. For example, although traces (<0.01%) of ethane occur at many levels, significant amounts of ethane only occur in the deeper coal beds. Also, the higher concentrations of ethane are associated with isotopically heavier methane (d13C values of about -55 to -35o/oo).
A vertical change sometimes takes place in the carbon isotopic composition (d13C) of the methane associated with the coalbed gas in the southern part of the Sydney basin (Figure 12). In boreholes 1, 6, and 12, the changes in d13C values of the methane with depth are minor, whereas in boreholes 3, 4, and 8, the changes are significant (about 15o/oo) with the shallower coals containing isotopically lighter methane than do the deeper coals. As demonstrated by Smith et al. (1985a), this isotopic change can not be accounted for by thermal maturity, because the calculated change in vitrinite reflectance over this depth interval (about 3 0 ft) is less than 0.05%.
Although Smith et al. (1985a) explained the depth-related changes by migration (desorption and diffusion), the author attributes the changes to alteration of the coalbed gases by mixing and oxidation in the shallower coals. The original, or unaltered, thermogenic gas occurs in the deeper beds, is the wettest, and contains the isotopically heaviest methane. At shallower depths, these thermogenic gases have probably degassed to a certain extent, have been mixed with or been replaced by late stage biogenic gas, and the heavier hydrocarbons might have undergone oxidation. The late-stage biogenic gas generation probably took place by carbon dioxide reduction as indicated by carbon dioxide data. In addition to the methane d13C values becoming more positive with depth, the concentration and carbon isotopic composition (d13C) of the carbon dioxide also change; in borehole 8 the concentration of carbon dioxide decreases from 22.7 to 1.8% and the d13C values become more positive (-4.8 to +11.1o/oo) with increasing depth (Smith et al., 1985a). With increasing depth and carbon dioxide reduction, the amount of carbon dioxide would be reduced and the isotopic composition (d13C) of the carbon dioxide substrate would become more positive because the methanogenes preferentially reduce the 12C-enriched carbon dioxide first (Claypool and Kaplan, 1974). As a consequence, both the carbon dioxide and resulting methane become isotopically heavier with depth.
Dai et al. (1987) studied the coalbed gases from coal mines and deep boreholes from several provinces in eastern China. The coals vary in age from Carboniferous to Tertiary and in rank from high-volatile C to anthracite (R0 values of 0.5 to 3.8%). They also analyzed the gases generated from a Tertiary coal in a pyrolysis experiment. Because of the variation in the age and location of the Chinese samples, it is difficult to determine the effects of coal composition on gas composition. However, these analyses form an excellent data set for observing the effects of secondary alteration on coalbed gas composition.
The coalbed gases are characterized by wetness (C2+) values between 0 and 34% and carbon dioxide contents between 0.1 and 5.1%. Figure 10 shows the good correlation between gas wetness (C2+) and both rank and depth. The wettest gases, particularly those from deep boreholes and the pyrolysis experiment of coal, occur in coals at ranks of high-volatile A to medium-volatile bituminous (R0 values from about 1.0 to 1.7%). However, coalbed gas samples from shallower mines are methane-rich in composition throughout their wide range of thermal maturity.
The methane d13C values from eastern China range from -66.9 to -24.9o/oo. As was also observed in western Germany, no correlation exists between methane d13C values and rank (R0), when all samples are considered (Figure 11A). However, the gas samples with the isotopically lightest methane generally occur at the shallowest depths where the gases are also methane-rich.
Dai et al. (1987) considered the coalbed gases from the deep borehole and pyrolysis experiment to be generally unaltered. These gases plot along their coalbed gas regression line shown in Figure 11. At a given R0, the methane from these Chinese coalbed gases has an intermediate d13C composition; it is several permil lighter than methane from unaltered German coalbed gases and several permil heavier than methane generated from type II/III kerogens (Figure 11A).
Although Dai et al. (1987) interpreted the depth changes in coalbed gas composition to be mainly the result of migration, the changes can best be explained by the mixing of late-stage biogenic gas and oxidation of wet gases in the zone of alteration. The zone of alteration was determined by Dai et al. (1987) to occur at different levels in different locations.
Zhang (1991) identified secondary or late-stage biogenic gas in coal beds in northeastern China. The identification of this gas was based on the occurrence of isotopically light biogenic methane (methane d13C values more negative than -55o/oo) with isotopically heavy thermogenic ethane (ethane d13C values more positive than about -35o/oo). This late-stage biogenic gas occurs in thermally mature (with respect to thermogenic hydrocarbon generation) coals. These coals experienced late Paleozoic uplift and erosion and there has been abundant time for alteration of the original coalbed gas at shallow depths.
The Lower Silesian basin of southwestern Poland is a major underground coal mining area characterized by frequent outbursts of gases and rocks, and gas explosions. The Upper Carboniferous Walbrzych and Zacler formations are the major coal-bearing sequences. Kotarba (1988, 1990) analyzed coal and associated gas samples from two areas within the Lower Silesian coal basin; these analyses represent the most extensive data set from underground mines. Kotarba collected both desorbed and "free" gases. The "free" gases were collected from 20 ft holes drilled into fresh coal surfaces and include gases both filling the cracks and degassed during drilling and sampling. The d13C values of both methane and carbon dioxide of the desorbed gases were always more positive by several permil as compared to the "free" gases.
The Upper Carboniferous coals vary in rank from high-volatile A bituminous to anthracite (R0 values between 1.1 and 2.6%) with rank increasing with depth (Kotarba, 1988, 1990). The coalification process, including the generation of hydrocarbons, was completed about 300 Ma (Westphalian time) at which time uplift and erosion took place. The coals contain abundant vitrinite (58 to 97%) and lesser amounts of liptinite (0 to 14%) and inertinite (3 to 40%).
The coalbed gases are highly variable in both their molecular and isotopic composition (Table 1) (Kotarba, 1988, 1990). Wetness (C2+) values range from 0.1 to 48.5%, whereas the carbon dioxide content ranges from 0.1 to 99.7%. The wettest gases are present where the carbon dioxide values are greater than 90%. In reference to isotopic composition, methane d13C values range from -66.1 to -24.6o/oo; methane dD values from -256 to -117o/oo; ethane d13C values from -27.8 to -22.8o/oo; and carbon dioxide d13C values from -26.6 to +16.8o/oo. A relatively good correlation appears to exist between methane d13C and methane dD values (Figure 7). In contrast, considerable scatter exists in plots between methane d13C versus ethane d13C values (Figure 8) and methane d13C values versus rank (R0) (Figure 11C).
Kotarba (1988, 1990) interpreted the coalbed gases to be mainly thermogenic. The gases with isotopically heavy methane (d13C values as much as -24.6o/oo) were probably modified by migration and/or regeneration similar to the isotopically heavy methane in western Germany and are summarized by regression line of Whiticar (1992) (Figure 11C). However, the chemically dry, isotopically light methane (d13C values more negative than -55 ppt) are interpreted by the author to be biogenic.
Kotarba (1988, 1990) attributed the lack of strong correlations between methane d13C values and rank (R0) (Figure 11) and between methane and ethane d13C values (Figure 8) to be the result of cracking of variable amounts of n-alkanes and migration. These relations are hereby interpreted to be primarily the result of mixing of biogenic gas and oxidation of heavier hydrocarbons in the zone of alteration. Late-stage biogenic gas generation is indicated by isotopically light biogenic methane being mixed with minor amounts of isotopically heavy thermogenic ethane (Figure 8). The zone of alteration is interpreted to extend to depths between 2600 and 3300 ft and to be controlled by seals, faults, and groundwater movement (Kotarba, 1988).
The largest gas and rock outbursts occur in areas with the highest concentrations of carbon dioxide. These high amounts of carbon dioxide are characterized by d13C values that vary from -5 to -8o/oo, with an average of about -7o/oo (Figure 16). This type of carbon dioxide is interpreted to be of magmatic and/or upper crust origin and to have migrated vertically along deep-seated faults (Kotarba, 1988, 1990). This migrating carbon dioxide preferentially expelled other hydrocarbon gases, particularly methane, resulting in the wettest gases in the basin. In contrast, the d13C of carbon dioxide occurring in lower concentrations is isotopically lighter (Figure 16) and has a different origin, such as generation during the coalification process.
San Juan Basin, New Mexico and Colorado, USA
The San Juan basin of northwestern New Mexico and southwestern Colorado (Figure 2) is a major gas-producing province in the United States with the main production coming from low-permeability Cretaceous sandstones. Recently, there has been active exploration for and production of gas from coal beds of the Upper Cretaceous Fruitland Formation; more coalbed gas has been produced to date from this basin than any other basin in the United States. The in-place gas resources in the Fruitland coal beds are estimated to be about 50 Tcf (ICF Resources, 1990).
Across the central basin, thermal maturity of the Fruitland coal increases to the northeast with R0 values ranging from about 0.4 to 1.5% (Figure 17), which equates to coal ranks of about subbituminous C to medium-volatile bituminous (Rice, 1983; Rice et al., 1989a; Law, 1992). Hydrocarbons (natural gas and some condensate) are produced from the Fruitland coal beds over most of this range of thermal maturity values. Maximum depths of burial for the Fruitland coal beds are about 4000 ft, but these depths do not coincide with the maximum levels of thermal maturity. Maximum depth of burial and present-day heat flow cannot account for the high maturity levels present at the north end of the basin (Figure 17). Law (1992) concluded that the high levels of thermal maturity in the northern part of the basin are the result of either (1) convective heat transfer associated with a deeply buried heat source located directly below the northern part of the basin, or (2) the circulation of relatively hot fluids into the basin from a heat source located in the vicinity of the San Juan Mountains to the north.
The Fruitland coals are composed mostly of vitrinite (>80%) and can be classified in a general way as humic (Rice et al., 1989a). Hydrogen indices from Rock-Eval pyrolysis are mostly between 200 and 400 at intermediate levels of thermal maturity and the atomic H:C ratios are between 0.8 and 1.2 (Rice et al., 1989a; Clayton et al., 1991; Michael et al., in press). These values are typical for types II and III kerogen dispersed in shales (Espitalie et al., 1977) and indicate a potential to generate wet gas and liquid hydrocarbons at intermediate ranks. In addition, examination of macerals in etched polished surfaces allows for the identification of hydrogen-rich matrix gel, a type of vitrinite (Rice et al., 1992; R.W. Stanton, personal communication, 1992) The hydrogen-rich matrix gels and liptinite macerals make up as much as 30% of the coal and indicate a potential for the coals to generate oil and wet gas.
Fruitland coalbed gas samples from a large number of producing wells display a variable composition with gas wetness (C2+) values ranging from 0 to 23% and carbon dioxide contents ranging from less than 1 to 13% (Scott et al., 1991). As shown on Figures 18 and 19, the producing part of the basin can be divided into two areas based on coalbed gas composition (Scott et al., 1991). In the south, the gases are generally wet (C2+ values generally greater than 6%) with minor amounts of carbon dioxide (generally less than 1%). Oil is produced in association with the wet gases in part of this area and the oils are described by Rice et al. (1989a), Clayton et al. (1991), and Clayton (this volume). In contrast, the gases in the north area are dry (C2+ values generally less than 3%), but contain significant amounts of carbon dioxide (generally greater than 6%). The boundary between these two areas is approximately parallel to the thermal maturity trends of the basin (Figure 17), and the change in composition between the two areas is rather abrupt. The composition and areal distribution of these produced gases are roughly equivalent to trends using the molecular composition of desorbed gas from Fruitland coal samples (Hanson, 1990).
Methane d13C values for the coalbed gases range from -46.6 to -38.5o/oo with the isotopically lightest methane restricted to the southwest part of the basin (Table 1) (Rice et al., 1989a). However, over most of the productive area and across the boundary between the two areas characterized by distinct molecular gas compositions, the methane d13C values display little variation and are in the range of -42 to -43o/oo (Table 1; Rice et al., 1989a). When plotted versus rank (R0) on Figure 11D, the methane d13C values form a relatively tight cluster that generally parallels the regression line for gases generated from types II/III kerogen.
The composition of the Fruitland coalbed gases is interpreted to be controlled by coal rank and composition, mixing, and oxidation. Based on the composition of the gases and the rank of the associated coal, the coalbed gases appear to be generally thermogenic in origin. However, at an R0 value of about 0.5% in the southern part of the basin, coalbed gases from the Fruitland are greater than 99% methane and the methane component is isotopically light (methane d13C value of -46.6 to -46.5o/oo) suggesting possible biogenic gas (Rice et al., 1989a). At R0 values of about 0.6 to 0.8%, wet gas and oil are produced from the coal beds; studies by Michael et al. (in press) indicate that a R0 value of 0.6% is the threshold for generation of thermogenic hydrocarbons from the Fruitland coals. The coal beds are considered to be the source of these thermogenic hydrocarbons because of the hydrogen-rich nature of the coals, as discussed earlier, and the desorption composition of the gases (Hanson, 1990). Methane d13C versus rank (R0) values for the coalbed gases fall along the regression line for mixed type II and III kerogen as defined by Faber (1987) (Figure 11D), further indicating the hydrogen-rich coals as the source of the gases. In addition, these wet gases are situated along the maturation pathway for a hydrogen-rich kerogen on an ethane d13C versus methane d13C plot (Figure 8).
With increasing rank (R0 values of 0.9 to 1.5%) and to the north, a gradual change to chemically drier gas with isotopically heavier methane would be expected because of the relatively consistent composition of the coals (Rice et al., 1989a; Clayton et al., 1991; Michael et al., in press). However, the change in the molecular composition to a methane-rich gas with significant amounts of carbon dioxide is rather abrupt (Figures 18 and 19), yet the methane d13C values are confined to a narrow range. This rapid change in molecular composition and insignificant change in isotopic composition are interpreted to be tied to the basin hydrology and microbial activity as described by Kaiser et al. (1991) a d Scott et al. (1991). The north area of distinct gas composition coincides with a region where the Fruitland coals and upper part of underlying Upper Cretaceous Pictured Cliffs Sandstone are defined as an aquifer with recharge occurring along the wet elevated area to the north. The boundary between the two gas types occurs along the southern limit of the aquifer resulting from the pinchout of the thick, continuous coals and/or displacement of coals along faults (Kaiser et al., 1991). The zone of alteration in the Fruitland, then, is developed over the entire north end of the basin and along the margins of the basin elsewhere.
Groundwater flow in the coal beds in this north area probably led to intensified microbial activity and alteration of the coalbed gas composition. The methane-rich composition of the gas in this north area can be explained by the aerobic bacterial consumption of the heavier gases (C2+), a process described by James and Burns (1984). Later anaerobic microbial activity in the aquifer also probably led to the generation of late-stage, isotopically light methane and isotopically heavy carbon dioxide (Carothers and Kharaka, 1980; Scott et al., 1991; Kaiser et al., 1991). The relatively uniform methane d13C values of the coalbed gas over a wide range of R0 values (0.9 to 1.5%) can be explaine by the mixing of late-stage, isotopically light biogenic methane with predominantly isotopically heavier thermogenic methane.
The d13C values of the high amounts of carbon dioxide in the north part of the basin are heavy (carbon dioxide d13C values of +6.8 to +18.6o/oo); Kaiser et al. (1991) also reported heavy d13C values (+16.7 to +26.0o/oo) for the total dissolved carbonate species (mainly bicarbonate) in the formation waters. These heavy carbon dioxide d13C values are evidence that late-stage biogenic gas generation by carbon dioxide reduction probably occurred. The reason for heavy d13C values is that methanogenic microbes preferentially reduce 12C carbon dioxide, resulting in the remaining substrate being enriched in 13C (Claypool and Kaplan, 1974).
The Piceance basin of northwest Colorado (Figure 2) is a hydrocarbon-rich basin with large estimated resources of coalbed gas and gas in low-permeability reservoirs of Cretaceous age, and oil shales of Tertiary age. The basin has large estimated coalbed gas resources (84 Tcf) (ICF Resources, 1990), which are contained in coal beds in the lower part of the Upper Cretaceous Mesaverde Group.
R0 values for the coals in the lower part of the Mesaverde increase regularly from less than 0.7% around the margins of the basin to greater than 2.1% along a trough in the southeastern part of the basin (Johnson and Rice, 1990). Present-day geothermal gradients increase from north to south across the basin. Paleo-geothermal gradients are interpreted to have been higher in the south as well, resulting from Tertiary igneous and volcanic activity associated with the Colorado mineral belt (Tweto, 1975).
Coals of the Mesaverde Group in the Piceance basin generally consist mainly of vitrinite (>75%) and lesser amounts of liptinite and inertinite (Collins, 1976; Law et al., 1989). Collins (1976) reported that coals in the lower part of the Mesaverde in the southeastern part of the Piceance basin have liptinite contents as high as 20%. In the central part of the basin, Rice et al. (1992) and R.W. Stanton (personal communication, 1992) reported higher amounts of liptinite and possibly hydrogen-rich vitrinite in the Mesaverde coals.
Coalbed gases from the Mesaverde are quite variable in both molecular and isotopic compositions over a R0 range of 0.5 to 1.9% (Table 1). Wetness (C2+) values range from 0.1 to 17.8%, carbon dioxide content varies from 0 to 25.4%, and methane d13C values range from -60.3 to -29.1o/oo. Where data are available, less variation is present in ethane d13C (-32.9 to -30.5o/oo) and carbon dioxide d13C (-7.1 to -7.0o/oo) ratios. As illustrated by Figure 11D, coalbed gases from the Piceance basin display more vertical spread in methane d13C values over a R0 range from 0.5 to 1.9% than those from any other U.S. basin.
The coalbed gas samples from the Piceance can generally be clustered into three groups based on composition (Figure 11D), geographic location, and origin. In the northwest part of the basin, isotopically light methane (d13C values of -60.3 to -53.5o/oo) are reported from a shallow well (<1300 ft) where the coals have R0 values in the range of 0.5 to 0.6% (Tremain and Toomey, 1983). These methane-rich gases are interpreted to be of biogenic origin.
The other two clusters of coalbed gas samples are from areas of production where gases are interpreted to be of thermogenic origin. One area is the White River dome located on the north edge of the basin where both wet gas and oil are produced from coal beds in the Mesaverde Group of high-volatile B bituminous rank (R0 value of about 0.7%) (Nuccio and Johnson, 1983). The oil is similar to that produced from Cretaceous coals in the San Juan basin (Rice et al., 1989a; Clayton et al., 1991; Clayton, this volume). In the White River dome, the wetness (C2+) of the gas varies from 11.5 to 17.8% and methane d13C values range from -44.1 to -40.0o/oo. The wetness of the gases suggests generation from hydrogen-rich macerals (either liptinite and/or hydrogen-rich vitrinite). Generation from these types of macerals is also indicated by the position of the gases along (1) the mixed type II/III kerogen regression line on the methane d13C-R0 plot (Figure 11D) and (2) the maturation pathway for oil-prone kerogen on the methane d13C-ethane d13C plot (Figure 8).
The wet gases on the White River dome are also associated with large amounts of carbon dioxide (11.5 to 25.4%), which are also present in gases in the adjacent sandstone reservoirs (Johnson and Rice, 1990). Both the concentration and isotopic composition (d13C values of -7.1 to -7.0o/oo) of the carbon dioxide are distinctly different from the carbon dioxide in the northern part of the San Juan basin (Table 1), where the carbon dioxide was interpreted to be of biogenic origin related to groundwater movement. In the White River dome, the carbon dioxide may have resulted from the thermal destruction of upper Paleozoic carbonates or from deep-seated igneous activity. The carbon dioxide, which is very mobile, migrated upward from the deeper horizons along numerous deep-seated faults typical of the dome (R.C. Johnson, personal communication, 1992).
In the central part of the basin, coalbed gas is produced at depths as much as 6500 ft from low-volatile bituminous coals (R0 values range from 1.5 to 1.9%) (Reinecke et al., 1991). This is the deepest commercial production of coalbed gas in the United States. The gases are fairly uniform in composition with wetness (C2+) values of 3.3 to 5.2% and methane d13C values of -35.2 to -34.5o/oo; carbon dioxide concentrations are less than 4%. These thermogenic coalbed gases were generated at high levels of thermal maturity and the presence of higher hydrocarbon gases indicates their generation from hydrogen-rich macerals, probably similar to those that generated the wet gas and oil at lower levels of thermal maturity at White River dome.
Unlike the San Juan basin, the zone of alteration in the Piceance basin is probably confined to the margins of the basin, as is the groundwater flow (Tyler et al., 1991). This restriction is the result of greater depth of burial and low permeability of the coals.
Powder River Basin, Montana and Wyoming, USA
The Powder River basin of northeastern Wyoming and southwestern Montana (Figure 2) contains large resources of coal (as much as 1.3 trillion short tons (Choate et al., 1984) in anomalously thick (as much as 200 ft) coal beds of the Paleocene Fort Union Formation. These coals are surface mined and form some of the most economically important deposits in the United States because of their occurrence in thick beds, depth of occurrence (generally less than 2500 ft), and low sulfur content (Law et al., 1991).
Because of the large resources of coal, the coal beds are also considered to contain large gas resources. Gas shows have been noted for years from the shallow coal beds and adjacent sandstones, and some economic recovery of the gas from both types of reservoirs, coal beds and sandstones, has taken place in the past few years (Law et al., 1991). The in-place gas resources in these thick coal beds are estimated to be as much as 39 Tcf (Choate et al., 1984).
The rank of the major Paleocene coal beds is relatively low over the entire basin, ranging from lignite to subbituminous C (R0 values of 0.3 to 0.4%) (Law et al., 1991). These are the lowest rank coals from which commercial gas production has been established in the United States. The levels of thermal maturity are also low in the underlying Upper Cretaceous shales indicating a relatively low geothermal gradient for the basin (Nuccio, 1990). Burial history reconstructions indicate that as much as 2000 ft of overburden was eroded over most of the basin about 10 Ma (Nuccio, 1990), so that the coals have never been buried deeper than about 4500 ft.
Natural gas desorbed and produced from the Paleocene coal beds and adjacent sandstones in the Powder River basin is composed mostly of methane with minor amounts of carbon dioxide at depths ranging from a few hundred to more than 1800 ft. Methane makes up more than 99% of the hydrocarbon fraction. Carbon dioxide is as much as 10% in the desorbed coalbed gas samples, less than 3% in the produced gas samples, and less than 1% in gas produced from adjacent sandstone reservoirs; the variable amounts of carbon dioxide are influenced by its reactivity and solubility.
The dominant methane component is enriched in the light isotope 12C (methane d13C values range from -56.5 to -53. 8o/oo) and depleted in deuterium (methane dD values range from -333 to-307o/oo) (Table 1). Both the coalbed gas composition and associated coal rank are very uniform throughout the basin. The gas in the adjacent sandstone reservoirs is almost identical in composition to that in the coal beds, indicating that the coals are the source for both. Based on the molecular and isotopic composition of the gas and the low rank of the coal, the gases are interpreted to be biogenic in origin and to have been generated via the methyl type fermentation pathway (Figure 7) (Whiticar et al., 1986). The dD values for surface and produced waters in the area of coalbed gas production range from -142 to -134o/oo (P.D. Jenden, personal communication, 1992). Using these water data, equations and fractionation factors from Jenden and Kaplan (1986) and Whiticar et al. (1986) suggest that more than 80% of the gas was derived by carbon dioxide reduction.
The big question about the biogenic gas in the Powder River basin is the timing of its generation. Biogenic gas was undoubtedly generated and accumulated shortly after deposition of the peat during a time of rapid subsidence and deposition. This early-stage gas was trapped by interbedded shales and compaction folds. However, some or all of this early-formed biogenic gas probably degassed from the low-rank coals following regional uplift and erosion about 10 Ma and earlier along the flanks as the basin was forming. This uplift and erosion also led to the establishment of a regional aquifer system in the thick coal beds; some wells completed in the coal beds yield as much as 1700 barrels of water per day (Tyler et al., 1991). Ground-water flow in the coal beds, at least along the basin' margins, led to microbial activity resulting in the generation of late-stage biogenic gas by carbon dioxide reduction, as suggested by the isotopic composition of the gases and associated waters. The groundwater in the Paleocene coal beds is a sodium bicarbonate type (Law et al., 1991; Van Voast, 1991), which, because of the lack of sulfate, is conducive to methanogenesis. However, the widespread generation in the basin of late-stage bacterial gas is uncertain because of the discontinuous nature of the coal beds (Flores, this volume). Therefore, the zone of alteration for coalbed gases in the Powder River basin may be limited to the margins of the basin or possibly extends across the entire basin.
Black Warrior Basin, Alabama and Mississippi, USA
The Black Warrior basin of Mississippi and Alabama (Figure 2), a southern extension of the Appalachian basin, is an important area for commercial coalbed gas production in conjunction with underground coal mining. The coal beds are assigned to the Lower and Upper Pennsylvanian (Upper Carboniferous) Pottsville Formation and their in-place gas resources are estimated to be about 20 Tcf (ICF Resources, 1990).
Coalbed gas production is so far restricted to the southeast part of the basin where coal beds range in rank from high-volatile A bituminous to low-volatile bituminous (R0 values of 0.8 to 1.4%) (Table 1) (Pashin, 1991). The coal beds occur at relatively shallow depths (<3000 ft); maximum depth of burial occurred in late Paleozoic time, which was followed by uplift and erosion in early Mesozoic time and subsequent deposition of Cretaceous strata. The high rank of the coals in the southwestern part of the basin, versus depth, is postulated to be related to upwelling hot waters along fractures (Winston, 1990). The average composition of the productive coals is 80.1% vitrinite, 15.3% inertinite, and 4.6% liptinite (R.B. Winston, personal communication, 1992). However, one o the coal zones averages 12.7% liptinite and contains as much as 19%, which indicates its ability to generate wet gas.
The produced coalbed gases are generally dry in molecular composition; wetness (C2+) values are generally about 0.1%, although the wetness of one sample was about 4%. In addition, the coalbed gases are characterized by variable methane d13C values of -51.0 to -41.9o/oo. Carbon dioxide contents are low (<1%).
The gases are interpreted to be thermogenic in origin, but altered by secondary processes, such as mixing of biogenic gas and oxidation of heavier hydrocarbons. If unaltered, these coalbed gases would be expected to plot near the regression lines of Dai et al. (1987) and Whiticar (1992) as shown on Figure 11D. On a methane d13C versus rank (R0) plot (Figure 11D), the coalbed gases from the Black Warrior basin form a relatively small group in which methane d13C values are lighter than the values predicted for generation from any of the types of kerogen. Alteration by secondary processes are favored by an active groundwater system and tectonically enhanced cleats (Pashin, 1991), in addition to shallow depth, and uplift and erosion. The extent of the zone of alteration for the coalbed gases beyond the area of present production is not known.
Coal beds have long been known to be source rocks for major resources of natural gas and liquid hydrocarbons. Recently, because of a tax credit given for the production of natural gas from coal beds in the United States, coal beds have become recognized as reservoirs for hydrocarbons, mainly natural gas. The molecular and isotopic composition of coalbed gases, that is, gases generated in and contained in coal beds, is highly variable, more so than gases generated from dispersed types I, II, or III kerogen. This variability reflects the multiple controls on their composition and origins.
The following conclusions can be made about the composition and origins of coalbed gas.
1. Large amounts of natural gas are generated in coal beds throughout their burial history. The gas is generated in two main ways: biogenic and thermogenic processes. Biogenic gas is generated by the degradation of the abundant organic matter in coal at shallow depths and low temperatures in rapidly accumulating sediments during early stages of coalification and at low ranks. Biogenic gas can also be generated in coal beds during later stages of geologic history, commonly after uplift and erosion and in association with groundwater flow. This late-stage generation can take place in coals of any rank. Thermogenic coalbed gas generation results from devolatilization of the coal beds, during which time large quantities of hydrocarbons, carbon dioxide, and water are liberated. Thermogenic generation of coalbed gas begins at a rank of about high-volatile C bituminous rank (R0 value of 0.6%) and increases with rank. The thermogenic gas can be generated directly from kerogen or by cracking of heavier hydrocarbons that are unable to migrate out of the coal.
2. Large quantities of natural gas are stored in coal beds on the internal surfaces of the organic matter (adsorption) or within the molecular structure of the coal (absorption). Natural gas migrates through the microstructure of the coal, by desorption and diffusion processes, due to the reduction of pressure which results from uplift and erosion, coal mining, and gas production from wells.
3. Coalbed gases are quite variable in their molecular and isotopic composition. In addition to methane, these gases can contain significant amounts of heavier hydrocarbon gases (C2+ values sometimes greater than 20%) and carbon dioxide (as much as 99%). Therefore, they should be referred to as coalbed gas and not coalbed methane. The primary controls of hydrocarbon coalbed gas composition are probably rank, composition, and depth/temperature of the associated coal. Biogenic gases are mostly methane, whereas thermogenic gases can be wet at intermediate ranks (high-volatile bituminous to medium volatile bituminous) and are dry at higher ranks. Methane and ethane d13C and methane dD values generally become more positive with increasing rank (R0). In addition, hydrogen-rich coals (high in liptinite and/or hydrogen-rich vitrinite) generate wetter gases at intermediate levels of rank and isotopically lighter methane, at equivalent levels of rank, than do oxygen-rich coals (mostly vitrinite).
4. A stronger relation exists between coalbed gas composition and depth, than rank. Shallow coal beds have a tendency to contain relatively dry gases with isotopically light (d13C) methane, as compared to those from deeper coal beds. This trend is interpreted to be the result of the original gases being altered by secondary processes, such as mixing of biogenic gas and oxidation of the wet gas components.
5. The zone of alteration of coalbed gases can occur at variable depths and is controlled by the physical nature of the coal beds, burial history, and groundwater flow. It commonly occurs at shallow depths (few hundred to thousands of feet) and along the margins of basins. However, the zone can extend across basins if proper conditions exist.
6. The significant nonhydrocarbon component of coalbed gases is carbon dioxide. Because of its reactivity and solubility in formation waters, the carbon dioxide generated during devolatilization is commonly not preserved in present-day coalbed gases. Significant amounts of present-day carbon dioxide can be the result of several processes not related to coalification, including recent micro-biological activity, thermal degradation of carbonates, and migration from magma chambers or the upper mantle.
7. Because of the complexity of controlling factors, both primary and secondary, coalbed gas composition is difficult to interpret and only general statements can be made. The best interpretation can be made by considering all geologic and geochemical data in relatively small study areas.
Alperin, M.J., Reeburgh, W.S., and Whiticar, M.J., 1988, Carbon and hydrogen isotope fractionation resulting from anaerobic methane oxidation: Global Geochemical Cycles, v. 2, p. 279-288.
Barker, J.F., and Fritz, P., 1981, The occurrence and origin of methane in some groundwater flow systems: Canadian Journal of Earth Sciences, v. 18, p. 1802-1816.
Bates, R.L., and Jackson, J.A., eds., 1987, Glossary of geology, 3rd Edition: Alexandria, Virginia, American Geological Institute, 788 p.
Berner, U., Faber, E., and Stahl, W., 1992, Mathematical simulation of the carbon isotopic fractionation between huminitic coals and related methane: Chemical Geology (Isotope Geoscience Section), v. 94, p. 315-319.
Bertrand, P., Behar, F., and Durand, B., 1986, Composition of potential oil from humic coals in relation to their petrographic nature: Organic Geochemistry, v. 1-3, p. 601-608.
Boreck, D.L., and Weaver, J.N., 1984, Coalbed methane study of the Anderson coal deposit, Johnson County, Wyoming--a preliminary report: U.S. Geological Survey Open-File Report 84-831, 16 p.
Boreham, C.J., and Powell, T.G., 1993, Petroleum source rock potential of coal and associated sediments, in AAPG Studies in Geology No. 38, Hydrocarbons from coal, p. 133-158.
Boyer, C.M., II, Kelefant, J.R., Kuuskraa, V.A., and Manger, K.C., 1990, Methane emissions from coal mining: issues and opportunities for reduction: U.S. Environmental Protection Agency, Air and Radiation (ANR-445), EPA/400/9-90/008, 86 p. plus appendices.
Carothers, W.W., and Kharaka, Y.K., 1980, Stable carbon isotopes of HCO3- in oil-field waters: implications for the origin of CO2: Geochimica et Cosmochimica Acta, v. 44, p. 323-332.
Choate, R., Johnson, C.A., and McCord, J.P., 1984, Geologic overview, coal deposits, and potential for methane recovery from coalbeds--Powder River basin, in Rightmire, C.T., Eddy, G.E., and Kirr, J.N., eds., Coalbed methane resources of the United States: American Association of Petroleum Geologists Studies in Geology Series 17, p. 335-351.
Chung, H.M., Gormly, J.R., and Squires, R.M., 1988, Origin of gaseous hydrocarbons in subsurface environments: theoretical considerations of carbon isotope distribution: Chemical Geology, v. 71, p. 97-103.
Claypool, G.E., and Kaplan, I.R., 1974, The origin and distribution of methane in marine sediments, in Natural gases in marine sediments: New York, Plenum Press, p. 99-139.
Clayton, C., 1991, Carbon isotope fractionation during natural gas generation from kerogen: Marine and Petroleum Geology, v. 8, p. 232-240.
Clayton, J.L., 1993, Composition of crude oils generated from coals and coaly organic matter in shales, in AAPG Studies in Geology No. 38, Hydrocarbons from coal, p. 185-202.
Clayton, J.L., Rice, D.D., and Michael, G.E., 1991, Oil-generating coals of the San Juan basin, New Mexico and Colorado, U.S.A.: Organic Geochemistry, v. 17, p. 735-742.
Close, J.C., and Erwin, T.M., 1989, Significance and determination of gas content data as related to coalbed methane reservoir evaluation and production implications: Proceedings of 1989 Coalbed Methane Symposium, p. 37-55.
Coleman, D.D., Risatti, J.B., and Schoell, M., 1981, Fractionation of carbon and hydrogen isotopes by methane-oxidizing bacteria: Geochimica et Cosmochimica Acta, v. 45, p. 1033-1037.
Coleman, D.D., Chao, L.L., and Riley, K.M., 1988, Microbial methane in the shallow Paleozoic sediments and glacial deposits of Illinois, U.S.A.: Chemical Geology, v. 71, p. 23-40.
Collins, B.A., 1976, Coal deposits of the Carbondale, Grand Hogback, and southern Danforth Hills coal fields, eastern Piceance basin, Colorado: Colorado School of Mines Quarterly, v. 17, no. 1, 138 p.
Colombo, U., Gazzarrini, F., Gonfiantini, R., Kneuper, G., Teichmuller, M., and Teichmuller, R., 1970, Carbon isotope study on methane from German coal deposits, in Hobson, G.D., and Speers, G.C., eds., Advances in organic geochemistry, 1966: Oxford, Pergamon Press, p. 1-26.
Dai, J., Qi, H., Song, Y., and Guan, D., 1987, Composition, carbon isotope characteristics and the origin of coal-bed gases in China and their implications: Scientia Sinica (Series B), v. 30, p. 1324-1337.
Espitalie, J., Laporte, J.L., Madec, M., Marquis, F., Leplat, P., Paulet, J., and Boutefeu, A., 1977, Methode rapide de caracterisation des roches meres, de leur potential petrolier et de leur degre d'evolution: Review of Institut Francais du Petrole, v. 32, p. 23-42.
Faber, E., 1987, Zur isotopengeochemie gasformiger Kohlenwasserstoffe: Erdol Erdgas Kohle, v. 103, p. 210-218.
Fuex, A.N., 1980, Experimental evidence against an appreciable isotopic fractionation of methane during migration, in Douglas, A.G., and Maxwell, J.R., eds., Advances in organic geochemistry, 1979: Oxford, Pergamon Press, p. 725-733.
Galimov, E.M., 1988, Sources and mechanisms of formation of gaseous hydrocarbons in sedimentary rocks: Chemical Geology, v. 71, p. 77-96.
Gould, K.W., Hargraves, A.J., and Smith, J.W., 1987, Variation in the composition of seam gases issuing from coal: Bulletin of Australasian Institute of Mining and Metallurgy, v. 292, p. 69-73.
Grossman, E.L., Coffman, B.K., Fritz, S.J., and Wada, H., 1989, Bacterial production of methane and its influence on ground-water chemistry in east-central Texas aquifers: Geology, v. 17, p. 495-499.
Hale, B.W., and Firth, C.H., 1988, Production history of the San Juan Unit No. 6 well, northern San Juan basin, New Mexico, in Fassett, J.E., ed., Geology and coal-bed methane resources of the northern San Juan basin, Colorado and New Mexico: Rocky Mountain Association of Geologists, p. 199-204.
Hanson, W.B., 1990, Chemistry of Western Interior USA coal-bed gases based upon desorption of subsurface coal samples (abs): American Association of Petroleum Geologists Bulletin, v. 74, p. 1326.
Hunt, J.M., 1979, Petroleum geochemistry and geology: San Francisco, W.H. Freeman and Co., 617 p.
Hunt, J.M., 1991, Generation of gas and oil from coal and other terrestrial organic matter: Organic Geochemistry, v. 17, p. 673-680.
Iannacchione, A.T., and Puglio, D.G., 1979, Geological association of coalbed gas and natural gas from the Hartshorne Formation in Haskell and LeFore Counties, Okla.: Presented at Ninth International Congress of Carboniferous Stratigraphy and Geology, May 19-26, 1979, Champaign-Urbana, Illinois, 18 p.
Iannacchione, A.T., Kertis, C.A., Houseknecht, D.W., and Perry, J.H., 1983, Problems facing coal mining and gas production in the Hartshorne coalbeds of the western Arkoma Basin, OK: U.S. Bureau of Mines Report of Investigations 8795, 25 p.
ICF Resources, 1990, The United States coalbed methane resource: Quarterly Review of Methane from Coal Seams Technology, v. 7, no. 3, p. 10-28.
James, A.T., 1983, Correlation of natural gas by use of carbon isotopic distribution between hydrocarbon components: American Association of Petroleum Geologists Bulletin, v. 67, p. 1176-1191.
James, A.T., 1990, Correlation of reservoired gases using the carbon isotopic compositions of wet gas components: American Association of Petroleum Geologists Bulletin, v. 74, p. 1441-1458.
James, A.T., and Burns, B.J., 1984, Microbial alteration of subsurface natural gas accumulations: American Association of Petroleum Geologists Bulletin, v. 68, p. 957-960.
Jenden, P.D., and Kaplan, I.R., 1986, Comparison of microbial gases from the Middle America Trench and Scripps Submarine Canyon: implications for the origin of natural gas: Applied Geochemistry, v. 1, p. 631-646.
Jenden, P.D., and Kaplan, I.R., 1989, Origin of natural gas in Sacramento basin, California: American Association of Petroleum Geologists Bulletin, v. 73, p. 431-453.
Jenden, P.D., Newell, K.D., Kaplan, I.R., and Watney, W.L., 1988, Composition and stable-isotope geochemistry of natural gases from Kansas, Midcontinent, U.S.A.: Chemical Geology, v. 71, p. 117-147.
Johnson, R.C., and Rice, D.D., 1990, Occurrence and geochemistry of natural gases, Piceance basin, northwest Colorado: American Association of Petroleum Geologists Bulletin, v. 74, p. 805-829.
Juntgen, H., and Karweil, J., 1966, Gasbildung und Gasspeicherung in Steinkohlenflozen, Part I and II: Erdol Kohle, Erdgas, Petrochem, v. 19, p. 251-258, 339-344.
Juntgen, H., and Klein, J., 1975, Entstehung von Erdgas gus kohligen Sedimenten.: Erdol, Kohle, Erdgas, Petrochem, Erganzungsband, v. 1, p. 52-69.
Kaiser, W.R., Swartz, T.E., and Hawkins, G.J., 1991, Hydrology of the Fruitland Formation, San Juan basin, in Geologic and hydrologic controls on the occurrence and producibility of coalbed methane, Fruitland Formation, San Juan basin: Gas Research Institiute, Topical Report (August 1987-July 1990), GRI -91/0072, p. 195-241.
Karweil, J., 1969, Aktuelle Probleme der Geochemie der Kohle, in Schenk, P.A., and Havernaar, I., eds., Advances in organic geochemisty, 1968: Oxford, Pergamon Press, p. 59-84.
Khorasani, G.K., 1987, Oil-prone coals of the Walloon Coal Measures, Surat Basin, Australia, in Scott, A.C., ed., Coal and coal-bearing sequences--recent advances: Geological Society Special Publication 32, p. 303-310.
Kim, A.G., and Douglas, L.J., 1972, Hydrocarbon gases produced in a simulated swamp environment: U.S. Bureau of Mines Report of Investigations 7690, 15 p.
Kotarba, M., 1988, Geochemical criteria for the origin of natural gases accumulated in the Upper Carboniferous coal-seam-bearing formations in Walbrzych Coal Basin (in Polish with English summary): Stanislaw Staszio Academy of Mining and Metallurgy Scientific Bulletin 1199, 119 p.
Kotarba, M., 1990, Isotopic geochemistry and habitat of the natural gases from the Upper Carboniferous Zacler coal-bearing formation in Nowa Ruda coal district (Lower Silesia, Poland), in Durand, B., and Behar, F., eds., Advances in organic geochemistry, 1989: Oxford, Pergamon Press, v. 1, p. 549-560.
Law, B.E., 1992, Thermal maturity patterns of Cretaceous and Tertiary rocks, San Juan basin, Colorado and New Mexico: Geological Society of America Bulletin, v. 104, p. 192-207.
Law, B.E., Nuccio, V.F., and Stanton, R.W., 1989, Evaluation of source-rock characteristics, thermal maturation, and pressure history of the Upper Cretaceous Cameo coal zone, Deep seam well, Piceance basin, Colorado: Proceedings of 1989 Coalbed Methane Symposium, p. 341-353.
Law, B.E., Rice, D.D., and Flores, R.M., 1991, Coalbed gas accumulations in the Paleocene Fort Union Formation, Powder River Basin, Wyoming, in Schwochow, S.D., Murray, D.K., and Fahy, M.F., eds., Coalbed methane of western North America: Rocky Mountain Association of Geologists, p. 179-190.
Levine, J.R., 1987, Influence of coal composition on the generation and retention of coalbed natural gas: Proceedings of 1987 Coalbed Methane Symposium, p. 15-18.
Levine, J.R., 1991, The impact of oil formed during coalification on generation and storage of natural gas in coalbed reservoir systems: Proceedings of 1991 Coalbed Methane Symposium, p. 307-315.
Liu, D., Fu, J., Qin, J., and Shi, X., 1987, Discovery of lipitobiolite in Suqiao area-on the discrimination of coal-generated gases and oils and their prospects: Chinese Journal of Geochemistry, v. 6, no. 4, p. 297-308.
Lu, S.-T., and Kaplan, I.R., 1990, Hydrocarbon-generating potential of humic coals from dry pyrolysis: American Association of Petroleum Geologists Bulletin, v. 74, p. 163-173.
Mukhopadhyay, P.K., and Hatcher, P.G., 1993, Composition of coal, in AAPG Studies in Geology No. 38, Hydrocarbons from coal, p. 79-118.
Meissner, F.F., 1984, Cretaceous and lower Tertiary coals as sources for gas accumulations in the Rocky Mountain area, in Woodward, J., Meissner, F.F., and Clayton, J.L., eds., Hydrocarbon source rocks of the greater Rocky Mountain region: Rocky Mountain Association of Geologists, p. 401-431.
Michael, G.E., Anders, D.E., and Law, B.E., in press, Geochemical evaluation of Upper Cretaceous Fruitland Formation coals with respect to thermal maturity, San Juan basin, New Mexico and Colorado: Organic Geochemistry.
Nuccio, V.F., 1990, Burial, thermal, and petroleum generation history of the Upper Cretaceous Steele Member of the Cody Shale (Shannon Sandstone bed horizon), Powder River basin, Wyoming: U.S. Geological Survey Bulletin 1917-A, 17 p.
Nuccio, V.F., and Johnson, R.C., 1983, Preliminary thermal maturity map of the Cameo-Fairfield or equivalent coal zone through the Piceance Creek basin, Colorado: U.S. Geological Survey Miscellaneous Field Investigations Map MF-1359.
Pashin, J.C., 1991, Regional analysis of the Black Creek-Cobb coalbed-methane target interval, Black Warrior basin, Alabama: Geological Survey of Alabama Bulletin 145, 127 p.
Phillips, T.L., and Cross, A.T., 1991, Paleobotany and paleoecology of coal, in Gluskoter, H.J., Rice, D.D., and Taylor, R.B., eds., Economic Geology, U.S.: Geological Society of America,The Geology of North America, v. P-2, p. 483-502.
Reinecke, K.M., Rice, D.D., and Johnson, R.C., 1991, Characteristics and development of fluvial sandstone and coalbed reservoirs of Upper Cretaceous Mesaverde Group, Grand Valley field, Garfield County, Colorado, in Schwochow, S.D., Murray, D.K., and Fahy, M.F., eds., Coalbed methane of western North America: Rocky Mountain Association of Geologists, p. 209-225.
Rice, D.D., 1983, Relation of natural gas composition to thermal maturity and source rock type in San Juan basin, northwestern New Mexico and southwestern Colorado: American Association of Petroleum Geologists Bulletin, v. 67, p. 1119-1218.
Rice, D.D., 1992, Controls, habitat, and resource potential of ancient bacterial gas in Vialy, R., ed., Bacterial gas: Editions Technip, p. 91-118.
Rice, D.D., and Claypool, G.E., 1981, Generation, accumulation, and resource potential of biogenic gas: American Association of Petroleum Geologists Bulletin, v. 65, p. 5-25.
Rice, D.D., and Flores, R.M., 1990, Coalbed methane potential of Tertiary coal beds and adjacent sandstone deposits, Powder River basin, Wyoming and Montana (abs): American Association of Petroleum Geologists Bulletin, v. 74, p. 1343.
Rice, D.D., and Flores, R.M., 1991, Controls of bacterial gas accumulations in thick Tertiary coal beds and adjacent channel sandstones, Powder River basin, Wyoming and Montana (abs): American Association of Petroleum Geologists Bulletin, v. 75, p. 661.
Rice, D.D., and Threlkeld, C.N., 1982, Occurrence and origin of natural gas in ground water, southern Weld County, Colorado: U.S. Geological Survey Open-File Report 82-496, 6 p.
Rice, D.D., Clayton, J.L., and Pawlewicz, M.J., 1989a, Characterization of coal-derived hydrocarbons and source-rock potential of coal beds, San Juan basin, New Mexico and Colorado, U.S.A.: International Journal of Coal Geology, v. 13, p. 597-626.
Rice, D.D., Epsman, M.L., and Mancini, E.A., 1989b, Origin of conventional and coalbed gases in the Black Warrior Region, Alabama (abs): Proceedings of 1989 Coalbed Methane Symposium, p. 321.
Rice, D.D., Clayton, J.L., Flores, R.M., Law, B.E., and Stanton, R.W., 1992, Some geologic controls of coalbed gas generation, accumulation, and production, western United States (abs): U.S. Geological Survey Circular 1074, p. 64.
Saxby, J.D., Bennett, A.J.R., Corcoran, J.F.., Lambert, D.E., and Riley, K.W., 1986, Petroleum generation: simulation over six years of hydrocarbon formation from torbanite and brown coal in a subsiding basin: Organic Geochemistry, v. 9, p. 69-81.
Saxby, J.D., and Shibaoka, M., 1986, Coal and coal macerals as source rocks for oil and gas: Applied Geochemistry, v. 1, p. 25-36.
Schoell, M., 1980, The hydrogen and carbon isotopic composition of methane from natural gases of various origins: Geochimica et Cosmochimica Acta, v. 44, p. 649-661.
Schoell, M., 1983, Genetic characterization of natural gases: American Association of Petroleum Geologists Bulletin, v. 67, p. 2225-2238.
Schoell, M., 1988, Multiple origins of methane in the Earth: Chemical Geology, v. 71, p. 1-10.
Scott, A.D., Kaiser, W.R., and Ayers, W.B., Jr., 1991, Composition, distribution, and origin of Fruitland Formation and Pictured Cliffs Sandstone gases, San Juan basin, Colorado and New Mexico, in Schwochow, S.D., Murray, D.K., and Fahy, M.F., eds., Coalbed methane of western North America: Rocky Mountain Association of Geologists, p. 93-108.
Shen, P., Shen, Q., Wang, X., and Xu, Y., 1988, Characteristics of isotope composition of gasiform hydrocarbon and identification of coal-type gas: Scientia Sinica (Series B), v. 31, no. 6, p. 735-74.
Smith, J.T., and Ehrenberg, S.N., 1989, Correlation of carbon dioxide abundance with temperature in clastic hydrocarbon reservoirs: relationship to inorganic chemical equilibrium: Marine and Petroleum Geology, v. 6, p. 129-135.
Smith, J.W., and Gould, K.W., 1980, An isotopic study of the role of carbon dioxide in outbursts in coal mines: Geochemical Journal, v. 14, p. 27-32.
Smith, J.W., Gould, K.W., Hart, G.H., and Rigby, D., 1985a, Isotopic studies of Australian natural and coal seam gas: Bulletin of Australasian Institute of Mining and Metallurgy, v. 290, p. 43-51.
Smith, J.W., Rigby, D., Gould, K.W., Hart, G., and Hargraves, A.J., 1985b, An isotopic study of hydrocarbon generation processes: Organic Geochemistry, v. 8, p. 341-347.
Stach, E., Mackowsky, M.-Th., Teichmuller, M., Taylor, G.H., Chandra, D., and Teichmuller, R., 1982, Stach's textbook of coal petrology: Berlin, Gebruder Borntraeger, 535 p.
Sundberg, K.R., and Bennett, C.R., 1983, Carbon isotopic paleothermometry of natural gas, in Bjoroy, M., ed., Advances in organic geochemisty, 1981: Chichester, John Wiley, p. 769-774.
Tang, Y., Jenden, P.D., and Teerman, S.C., 1991, Thermogenic methane formation in low-rank coals-published models and results from laboratory pyrolysis of lignite, in Manning, D.A.C, ed., Organic geochemistry--advances and applications in the natural environment: Manchester, Manchester University Press, p. 329-331.
Thomas, J., Jr., and Damberger, H.H., 1976, Internal surface area, moisture content, and porosity in Illinois coals: variations with coal rank: Illinois Geological Survey Circular 493, 38 p.
Tissot, B.P., and Welte, D.H., 1984, Petroleum formation and occurrence: New York, Springler-Verlag, 699 p.
Tremain, C.M., and Toomey, J., 1983, Coal bed methane desorption data: Colorado Geological Survey Open-File Report 81-4, 514 p.
Tweto, O., 1975, Laramide (Late Cretaceous-early Tertiary) orogeny in the southern Rocky Mountains: Geological Society of America Memoir 144, p. 1-44.
Tyler, R., Ambrose, W.A., Scott, A.R., and Kaiser, W.R., 1991, Coalbed methane potential of the Greater Green River, Piceance, Powder River, and Raton basins: Gas Research Institute, Topical Report (January 1991-July 1991), GRI-91/0315, 244 p.
U.S. Geological Survey and Minerals Management Service, 1989, Estimates of undiscovered conventional oil and gas resources in the United States; A part of the nation's energy endowment: U.S. Department of Energy, 44p.
van Krevelen, D.W., 1981, Coal (typlogy, chemistry, physics, and constitution): Amsterdam, Elsevier, 514 p.
Van Voast, W.A., 1991, Hydrogeologic aspects of coal-bed methane occurrence, Powder River basin (abs): American Association of Petroleum Geologists Bulletin, v. 75, p. 1142-1143.
Welte, D.H., Schaefer, R.G., Stoessinger, W., and Radke, M., 1984, Gas generation and migration in the Deep basin of western Canada: American Association of Petroleum Geologists Memoir 38, p. 35-47.
Whiticar, M.J., 1992, Stable isotope geochemistry of coals, humic kerogens, and related natural gases: Proceedings of the Canadian Coal and Coalbed Methane Geoscience Forum, B. C. Geological Survey Branch, Alberta Geological Survey, and Geological Survey of Canada, p. 149-172.
Whiticar, M.J., and Faber, E., 1986, Methane oxidation in sediment and water column environments--isotope evidence: Organic Geochemistry, v. 10, p. 759-768.
Whiticar, M.J., Faber, E., and Schoell, M., 1986, Biogenic methane formation in marine and freshwater environments, CO2 reduction vs. acetate fermentation--isotopic evidence: Geochimica et Cosmochimica Acta, v. 50, p. 693-709.
Winston, R.B., 1990, Preliminary report on coal quality trends in Upper Pottsville Formation coal groups and their relationships to coal resource development, coalbed methane occurrence, and geologic history in the Warrior coal basin, Alabama: Geological Survey of Alabama Circular 152, 53 p.
Woese, C.R., Kandler, O., and Wheelis, M.L., 1990, Towards a natural system of organisms: proposal for the domains Archaea, Bacteria, and Eucarya: Proceedings of the National Academy of Science (U.S.A.), v. 87, p. 4576-4579.
Woltemate, I., Whiticar, M.J., and Schoell, M., 1984, Carbon and hydrogen isotopic composition of bacterial methane in a shallow freshwater lake: Limnology and Oceanography, v. 29, p. 985-992.
Wyman, R.E., 1984, Gas resources in Elmworth coal seams, in Masters, J.A., ed., Elmworth-case study of a deep basin gas field: American Association of Petroleum Geologists Memoir 38, p. 173-188.
Zhang, Y.G., 1991, Generation, accumulation, and preservation of natural gas (in Chinese): Nanjing, P.R. China, River and Sea University Press, 176 p.
Zhang, Y.G., and Chen, H.J., 1985, Concepts on the generation and accumulation of biogenic gas: Journal of Petroleum Geology, v. 8, p. 405-422.
I thank C. N. Threlkeld and A. Warden of the U. S. Geological Survey for molecular and isotopic analyses of the natural gases reported for the first time in this paper. I also acknowledge the cooperation and assistance of the companies that assisted in our sampling program. The author benefited from discussions with and/or reviews by George E. Claypool, Mobil Exploration and Producing Technical Center; William B. Hanson, Amoco Production Co.; Joseph R. Hatch, U. S. Geological Survey; John M. Hunt, Woods Hole Oceanographic Institute; Peter D. Jenden, Chevron Oil Field Research Company; Maciej Kotarba, Polish Academy of Mining and Metallurgy; J. Bruno Risatti, Illinois State Geological Survey; and Yi-Gang Zhang of Chinese Ministry of Geology and Mineral Resources. Thanks are extended to William B. Hanson of Amoco Production Co. and Richard E. Wyman of Canadian Hunter Exploration Ltd. for data on coalbed gas compositions.
