![]() Click to view page images in PDF format.
Click to view page images in PDF format.
Microporosity in Arab Formation Carbonates, Saudi Arabia*
By
Dave L. Cantrell1 and Royal M. Hagerty2
Search and Discovery Article #30008 (2002)
*Adapted for online presentation from article with the same title by the same authors published in GeoArabia, Vol. 4, No. 2, 1999, Gulf PetroLink, Bahrain (www.gulfpetrolink.com.bh). Appreciation is extended to the authors and to Moujahed Al-Husseini, Gulf Petrolink, Editor-in-Chief of GeoArabia ([email protected]).
1Saudi Aramco, Dhahran, Saudi Arabia ([email protected]).
2previously Exxon Production Research Company.
Microporosity
occurs throughout Arab Formation carbonates of Saudi Arabia, and affects the log
response, fluid flow properties and ultimate recovery of hydrocarbons in these
reservoirs. Qualitative examination of Arab samples indicates that microporosity
occurs as four major types: (1) microporous grains, (2) microporous matrix, (3)
microporous fibrous to bladed cements, and (4) microporous equant cements.
Quantitative estimation of microporosity abundance was measured in two ways: (1)
thin section point counts, and (2)  pore
pore throat size distributions derived from
capillary pressure data. Point count data shows that microporosity can vary
widely from sample to sample, ranging from 0% to 100% of the total measured
porosity of a sample. Capillary pressure data confirms the volumetric
significance of
throat size distributions derived from
capillary pressure data. Point count data shows that microporosity can vary
widely from sample to sample, ranging from 0% to 100% of the total measured
porosity of a sample. Capillary pressure data confirms the volumetric
significance of  pore
pore throats that are 10 microns or less in size. Variations in
microporosity abundance and type appear to be controlled by depositional
texture, grain mineralogy and grain microstructure. We suggest that
microporosity in Arab Formation carbonates formed diagenetically, via three
mechanisms: (1) leaching and incomplete reprecipitation of metastable carbonate,
(2) crystal growth contact inhibition, and (3) (locally) endolithic borings of grains.
throats that are 10 microns or less in size. Variations in
microporosity abundance and type appear to be controlled by depositional
texture, grain mineralogy and grain microstructure. We suggest that
microporosity in Arab Formation carbonates formed diagenetically, via three
mechanisms: (1) leaching and incomplete reprecipitation of metastable carbonate,
(2) crystal growth contact inhibition, and (3) (locally) endolithic borings of grains.
|
tCrystal growth contact inhibition
tCrystal growth contact inhibition
tCrystal growth contact inhibition
tCrystal growth contact inhibition
tCrystal growth contact inhibition
tCrystal growth contact inhibition
tCrystal growth contact inhibition
tCrystal growth contact inhibition
tCrystal growth contact inhibition
tCrystal growth contact inhibition
tCrystal growth contact inhibition
tCrystal growth contact inhibition
tCrystal growth contact inhibition
tCrystal growth contact inhibition
|
FIGURE/TABLE CAPTIONS (** in abbreviated form)
**Complete caption appears with full-sized image and after References.
Microporosity occurs in both sandstone and carbonate reservoirs and affects their fluid flow properties (i.e., productivity) as well as their log responses. In carbonate reservoirs in particular, the presence of micropores increases the capillary attraction to the wetting phase (typically water) and frequently results in abundant bound water in the micropores. Log calculations may then yield high water saturations and cause erroneous hydrocarbon estimates (Kieke and Hartmann, 1974; Keith and Pittman, 1983). Based on log calculations alone, one might not identify a reservoir that would flow little, if any, water. For this reason, it is necessary to consider microporosity in any comprehensive formation evaluation program. This study
documents the occurrence, distribution, abundance and origin of
significant amounts of microporosity in Arab Formation carbonates of
Saudi Arabia. Information for this study comes from microscopic
examination of petrographic thin sections and scanning electron
microscope (SEM) examination of fractured rock surfaces and epoxy This study uses a three-part approach: (1) microporosity is examined qualitatively throughout the Arab Formation, (2) variations in microporosity abundance in the Arab-D are discussed, and (3) a discussion of several possible mechanisms that could have caused microporosity to form in the Arab-D is included.
The Upper Jurassic Arab Formation (Tithonian and Kimmeridgian in age, see Al-Husseini (1997) for a discussion) consists of four geographically widespread carbonate-evaporite cycles or members. These members are labeled, from top down, Arab-A, -B, -C and -D (Figure 1a). The Arab-D Member is the oldest (deepest) and thickest of these cycles, and is generally interpreted to represent deposition in shallow shelfal to sabkha settings. The Arab-D consists of several (at least four) major upward-shoaling cycles that were initiated in deeper subtidal water and shoaled to near sea-level. These upwardshoaling cycles comprise a variety of skeletal grainstones and packstones with ooid grainstones locally common in the uppermost Arab-D. The top of the Arab-D carbonate is characterized by thin subtidal to intertidal carbonates with sabkha and subaqueous evaporites above. Most of the quantitative data used in this study comes from the Arab-D Member of several large fields in Eastern Saudi Arabia (Figure 1b). For a more complete treatment of the rock types, depositional environments and diagenesis of the Arab-D, see Mitchell et al. (1988) and Meyer and Price (1992). Many different
definitions for microporosity have been proposed in the literature.
Choquette and Pray (1970) defined a micropore as a For this study, we have defined microporosity to be all pores that are approximately 10 microns in diameter or smaller. This definition is empirical and is based on the idea that microporosity is the difference between total measured porosity and porosity visible from microscopic examination of thin sections (point count porosity). Since the optical resolution limit of most petrographic microscopes is approximately 10 microns, by default microporosity includes all pores less than 10 microns in size. As will be discussed later, capillary pressure data reveal that micropores much smaller than 10 microns (as small as 0.01 microns in diameter) are common in most Arab carbonates, although none of these very small micropores are optically resolvable with a standard petrographic microscope.
QUALITATIVE EXAMINATION OF MICROPOROSITY The first steps in understanding microporosity were to catalogue variations in types of microporosity and to document their occurrence. This study recognizes four major types of microporosity, namely: microporous grains, microporous matrix, microporous fibrous to bladed cements and microporous equant cements. Figure 2 schematically illustrates these four important micropore types. Of these, microporous grains and microporous matrix are the most volumetrically significant micropore types in the Arab Formation.
Microporous
grains are common throughout the Arab carbonates (Figure
3) and
constitute the most volumetrically significant microporosity type. SEM
examination of The internal
fabric of microporous grains consists of uniform-sized, subhedral
crystals of micrite ranging from 1 to 4 microns. SEM examination of SEM examination is not always necessary to recognize microporous grains, since both a blue hazy appearance in thin section and complete grain micritization usually indicate the presence of microporosity (Figures 3a; 4a and 4b; 5c). This blue hazy appearance in thin section results from the infiltration of blue-dyed epoxy into micropores in grains and matrix (lime-mud or micrite). Also, since microporous grains are composed of micrite crystals, complete grain micritization usually reflects the presence of microporosity (for example, compare the non-micritized and non-microporous cement crystal with the microporous micritized grain in Figure 3f). Microporous
matrix consists of 1 to 10 micron, subhedral calcite crystals that are
randomly packed to yield a continuous network of micropores that are
connected by smaller (1 micron maximum)
Microporous Fibrous to Bladed Cements Microporous
fibrous to bladed cements are common in Arab grainstones that contain
significant amounts of early marine, isopachous calcite cement. The
crystals of calcite cement are approximately 30 to 40 microns long and 5
to 10 microns wide (Figures 6e and 6f). SEM examination of epoxy Although these “intercrystalline” pores are volumetrically insignificant in the Arab-D, they are important in that they provide a pathway connecting microporous grains to one another and to macropores. These cements isopachously coat and connect microporous grains, and separate microporous grains from larger interparticle pores. Micropores in these cements thus could play an important role in communication between microporous grains and macropores.
Microporous
equant cements are less common than the microporous fibrous to bladed
cements described above, primarily because sparry equant calcite cement
itself is not as common in Arab-D reservoir rocks in Eastern Saudi
Arabia. Equant cements are composed of subhedral to anhedral crystals of
calcite approximately 30 to 40 microns in size (Figure
7c). These
micropores occur between cement crystals; similar to the microporous
fibrous to bladed cements discussed previously, they probably result
from the contact inhibition of adjacent growing crystals (Wardlaw,
1976).
QUANTITATIVE ASPECTS OF MICROPOROSITY The next aspect of this study was to quantitatively evaluate the abundance of microporosity, and this was accomplished in two ways. First, thin section point counts were used to determine microporosity abundance and variability; and second, mercury injection capillary pressure analysis allowed the resolution of greater details in the quantitative analysis of microporosity abundance and distribution in Arab-D samples. Petrographic Thin Sections Point Counts Petrographic thin sections were point counted to determine optically visible porosity. This visible porosity includes all pores greater than about 10 microns in size (in other words, macropores only). Visible porosity was then compared to the total porosity measured on the equivalent core plug (the thin section was cut from the clipped end of this core plug) by routine core analysis, to derive an estimate of the amount of microporosity in the sample. Sixty-three samples were analyzed in this manner. Figure 8 illustrates the relationship between measured and point count porosity values for the samples used in this study, as well as for 8 additional (non-Arab-D) calibration samples. For the Arab-D samples (open circles), measured (total) porosity is consistently higher than point count estimated porosity, with a linear regression of Y = 0.68X - 1.37. This plot also shows data for 8 additional calibration samples (red circles) that are included to validate the point count method for porosity determination. These additional samples all have minimal microporosity, yield similar measured porosity and point count porosity values, and tend to fall along a line described by Y = X. Overall, the
petrographic data indicate that microporosity occurs almost universally
throughout the Arab Formation (Figures 9 and
10). The abundance of
microporosity, however, varies from sample to sample and can account for
anywhere from 0% to 100% of the total measured porosity (øT). In individual samples, both solid non-microporous grains and
extremely leached, moldic-appearing grains are seen. Figure 9
illustrates microporosity abundance and variability, and indicates that
virtually all samples have a significant amount of microporosity. Even
the best reservoir quality rocks typically have 25% to 50% microporosity
(that is, 25% to 50% of the total While some microporosity typically occurs in all studied samples, the type and amount of microporosity varies systematically. The petrographic data allowed us to identify the three major factors that exercise some control over variations in microporosity abundance and variability: (1) depositional texture, (2) original mineralogy, and (3) grain microstructure.
The primary control over microporosity in a rock sample as a whole is depositional texture. This relationship between depositional texture and microporosity abundance (Figure 10) occurs principally because the overall proportion of microporous components (including microporous grains and microporous matrix) increases as depositional texture becomes muddier. For example, grainstones and mud-lean packstones consist predominantly of grains (which are usually microporous) and large macropores; relatively minor microporous matrix is present. In these rocks, then, microporosity is limited to microporous grains. Some microporous cements and matrix may be present, but they generally do not contribute much quantitatively to overall microporosity. Consequently, microporosity in these rock types is relatively low, averaging 19.8% and 37.6% of the total porosity (øT) for grainstones and mud-lean packstones, respectively. In muddier carbonates, however, both microporous grains and matrix are present, and macroporosity is less abundant. In these rocks, primary interparticle porosity is filled by microporous matrix, reducing macroporosity yet at the same time increasing the proportion of microporosity. Average microporosity for packstones is 53.8% of øT. In the case of mud-supported rocks (wackestones and mudstones), microporous matrix is dominant. In many cases, no visible macroporosity is present and microporosity accounts for the total porosity of the rock. Essentially 100% of the total porosity in wackestones and mudstones is microporosity. In general,
then, the proportion of total porosity consisting of microporosity
increases as depositional texture becomes muddier, due to filling of
Microporosity occurs only in grains originally composed of metastable aragonite or high magnesium calcite, whereas grains originally composed of low magnesium calcite are solid and non-microporous. Table 1 summarizes the interpreted primary mineralogies of common grains and cements in the Arab-D. This occurs without regard as to whether grains are skeletal or non-skeletal in origin. Different grain types with different original internal fabrics (e.g., ooids versus foraminifera) may be transformed into microporous grains with similar end-member fabrics (Figures 4a to 4e; 5a to 5e; 11a to 11c). Aragonite and high magnesium calcite are less stable than low magnesium calcite at conditions typical of the earth’s crust (Bathurst, 1975). This difference in stability is responsible for the formation of moldic porosity in aragonite grains and microporosity in most high magnesium calcite grains. As shown by the leaching of aragonitic molluscs, corals and dasycladacean algae, most aragonitic grains in the Arab-D are completely leached; high magnesium calcite grains remain but usually are microporous. In contrast, grains with presumed original mineralogy of low magnesium calcite (brachiopods, ostracodes, and stromatoporoids) never appear leached or microporous. Primary mineralogy thus controls the potential for complete micritization and microporosity development within a grain.
One factor that seems to modify the effect of primary mineralogy on microporosity formation is grain microstructure. Certain grains, specifically echinoderm fragments and early marine cement crystals, are not micritized or microporous, in spite of probably being originally composed of metastable high magnesium calcite (Figure 11c). These grains all have single-crystal microstructure and, except for rare micrite envelopes caused by boring activities of endolithic algae or fungi, are devoid of complete grain micritization and microporosity. While the role of microstructure is not completely understood, large, single-crystal grains (e.g., echinoderms) are probably more stable and less susceptible to dissolution and microporosity formation during diagenesis than fibrous or aggregate-crystalline grains. Fyfe and Bischoff (1965) have shown that very small grains (one micron and smaller) have solubilities that are significantly higher than those of macrocrystalline grains of the same material. Thus, the single-crystal microstructure is a more stable structure and probably inhibits to some degree the formation of diagenetic microporosity within a grain of metastable mineralogy.
Capillary
pressure data was also used to quantitatively evaluate microporosity in
Arab Formation carbonates. For this method, Mercury
injection data is a measure of D = 0.58(s)(cosq) where: D = P = Capillary pressure (mercury injection pressure) in psi s = Interfacial tension, dynes/cm (s = 485 dynes/cm for air/mercury system) q = Contact angle in degrees (q = 130° for air/mercury)
The F(D) = V x 104/DD where: F(D) = Distribution function (as a function of diameter) V = Incremental
volume of mercury injected (fraction of total DD = Incremental change in diameter over which V was determined While the percent of microporosity derived from capillary pressure data is not strictly comparable with microporosity determinations from thin section point counts, the capillary pressure data does allow a qualitative comparison of microporosity determined by two very different techniques. Results from
some of the capillary pressure analyses are shown in Figures 12 and
13,
and illustrate the complex Figure 13 also
illustrates how microporosity – and total porosity - vary with matrix
content and depositional
texture. As matrix content increases and depositional texture becomes
muddier, larger In general, the
capillary pressure data allows us to resolve quantitatively the actual
size range and distribution of microporosity. While the
This study has shown how microporosity was identified in Arab Formation carbonates and how its occurrence was related primarily to depositional texture and original mineralogy. This relationship can be extended further and used to predict and map out the spatial distribution of microporosity across a reservoir. This could be done by first constructing maps and cross-sections that show depositional texture variations (using core control), and then, using the microporosity-depositional texture relationship established by this study, predict microporosity amounts and distribution trends across a reservoir. These maps could be effective tools to predict and explain fluid flow, wireline log response and reservoir performance. It is important
to recognize that microporosity can have a significant impact on the
fluid flow properties and log response of a reservoir. Typically, water
is thought to be the wetting phase in a rock and, consequently,
capillary forces tend to draw water up into the very small pores (micropores).
Water held in these micropores by capillary attraction tends to be
“bound” (immobile) and may not be producible. Wireline logs record
total fluid content of a rock - including bound water in the micropores
- thus the resulting calculated water saturations do not reflect the
true producible fluid saturations that occur in the macropore system.
When no microporosity is present, wireline logs yield fluid saturations
that occur in the macropore system and are much more indicative of the
true producibility of the reservoir. Zones with significant water-filled
microporosity may contain (and produce) water-free hydrocarbons from the
macropore system, even though the logs may indicate the zone to be
apparently wet. Changes in rock wettability and Microporosity
can also have a significant impact on a reservoir performance model, for
two reasons. First, the actual effective porosity of the reservoir may
be significantly less than the total porosity indicated by wireline logs
and by core analysis data. Consequently, volumetric calculations using
these This study has shown how significant microporosity is, how it varies, and what are some of the controls over its occurrence in Arab Formation carbonates of the Middle East. However, we feel that the techniques developed during this study are applicable to other fields elsewhere, in that they represent a reasonable rapid and cost effective technique for documenting and quantifying microporosity variations within a given reservoir interval.
As Pittman (1971) observed, it is unlikely that any microporosity in ancient rocks can be termed primary, as any primary microporosity associated with metastable aragonite or high magnesium calcite phases must be modified substantially (if not destroyed) during inversion to low magnesium calcite. In a similar manner, most micropores in the Arab-D are probably diagenetic in origin, or at least have been modified somewhat by diagenesis. As previously noted, microporosity occurs in a variety of forms or types in the Arab-D, implying that several mechanisms are responsible for the formation of microporosity. In the following discussion, similar types of microporosity are treated together, since micropores with similar morphologies probably formed in the same manner. We should note that this discussion applies primarily to possible microporosity-forming mechanisms in the Arab-D Member; while microporosity has also been recognized in other Cretaceous carbonate reservoirs in the area (e.g. Budd, 1989; Moshier, 1989), we have not studied these other units and so conclusions from our study may not be appropriate for them. Three mechanisms are responsible for the majority of microporosity in the Arab-D: (1) leaching and incomplete reprecipitation of metastable carbonate, (2) crystal growth contact inhibition, and (3) endolithic boring of grains (locally).
Leaching and Incomplete Reprecipitation of Metastable Carbonate Post-depositional leaching of metastable CaCO3 minerals and incomplete reprecipitation of low magnesium calcite is probably responsible for microporosity in micritized microporous grains and matrix. Although grain micritization by boring endolithic algae and fungi is recognized as a significant grain-altering process in modern-day shallow water carbonate environments (Logan, 1974; Kobluk and Risk, 1977) and occurs locally in the Arab-D, several factors argue against an algal/fungal origin for most of the pervasive micritization and microporosity development in the Arab-D. First, complete micritization is mineralogy-selective in the Arab-D, occurring only in originally metastable components (Figures 11a to 11c). Boring endolithic organisms, however, would not be expected to display any preference in mineralogy of the host grains. Also, the extent of grain micritization is too pervasive in Arab carbonates to be attributed solely to algal or fungal activity. Although some micrite envelopes and borings of algal or fungal origin do occur, they are readily distinguishable from the completely micritized and microporous grains that are so common in these rocks (Figure 11c). Finally, the habit of Recent micropores of algal or fungal origin are significantly different from those seen in the Arab-D (Figures 11d to 11f). It appears that the pervasive micritization and microporosity development results from partial post-depositional solution of metastable CaCO3 minerals. Since the stabilization of metastable carbonate probably occurs relatively early (either during early marine or early fresh water diagenesis (Bathurst, 1975; Longman, 1980; Palmer et al., 1988)), the development of microporous grains and matrix was probably a relatively early diagenetic process. Also, since the appearance of microporosity is dependent on original mineralogy and microstructure, depositional facies controls the architecture of microporosity development. It is the mineralogy and microstructure of the grains typically comprising each facies that determines the appearance and abundance of microporosity present. Depositional facies thus forms the basis for predicting microporosity distribution. All of the rock-types found in the Arab-D in Saudi Arabia contain significant amounts of microporosity; however, the exact amount of microporosity in each rock-type depends on the proportion of grains within each that were originally composed of metastable mineralogy. There are several diagenetic processes under which the dissolution-incomplete reprecipitation process responsible for most Arab-D microporosity may have occurred. These include: (1) evaporite diagenesis, (2) leaching by carbon dioxide-charged fluids formed during the maturation and migration of hydrocarbons, (3) marine diagenesis, (4) fresh water vadose diagenesis, (5) fresh water phreatic diagenesis, and (6) mixing zone diagenesis. As we will explore in this section, we feel that evaporite diagenesis is probably the most likely process responsible for forming most of the microporosity in the Arab-D.
The presence of a thick evaporite sequence immediately overlying the Arab-D Reservoir influences the diagenesis of Arab-D carbonates. Previous workers (Broomhall and Allan, 1985; Wilson, 1985) have suggested a direct relationship between some of the Arab dolomites and the evaporites. Fluids expelled during the compaction of evaporites or during the transformation of gypsum to anhydrite with burial are usually undersaturated with respect to Ca+2 (Shearman, 1978). Other workers have shown that such fluids typically have high Mg+2/Ca+2 ratios and are capable of dolomitizing limestones (Butler, 1969; Clark, 1980; Moore and Druckman, 1981). Also, these fluids become fairly acidic (pH<5.5) at shallow burial depths (Butler, 1969). Other workers
concerned with the diagenesis of evaporite-carbonate sequences have
pointed out the role of evaporite-derived
Carbon Dioxide-Charged Subsurface Waters Carbonic acid
may be generated in
Diagenesis in the marine environment is a common process in many shallow-marine carbonate depositional environments. Typically, products of this environment include cementation, partial grain micritization (micrite envelope formation), some forms of dolomitization and, potentially, grain leaching. This last process is particularly significant in Upper Jurassic sea water. It is thought to have generally been “calcite facilitative” (Wilkerson et al., 1985), and capable of dissolving originally aragonitic grain components. Indeed, recent workers have described the occurrence of aragonite dissolution within the “marine-burial” diagenetic environment of shallow water Neogene carbonates in South Florida and the Bahamas (Melim and Swart, 1998). Such dissolution could have produced moldic porosity in originally aragonitic grains. However, partial dissolution of Neogene originally calcitic grains (to form microporosity) has not been observed, and it is likely that Jurassic originally calcitic grains would have been in equilibrium with sea water and would not have been significantly affected (dissolved) by continued immersion in sea water. Partial dissolution of these originally calcitic grains by sea water during marine diagenesis was probably not responsible for the microporosity observed in the Arab-D.
It seems likely
that local small islands could have developed upon subaerially exposed,
grain-rich, shoaling sands that reached sea-level during deposition of
the Arab-D. This is commonly seen in many regressive carbonate
Fresh Water Phreatic Diagenesis The geometry and extent of the fresh water phreatic zone is normally a function of topography, rainfall and the distribution of permeable pathways. In light of the probable low topography and arid climate that prevailed during Arab carbonate deposition (Mitchell et al., 1988), it seems unlikely that fresh water lenses of a sufficient size and hydrostatic head could have persisted. This view is supported by the obervation that interpreted fresh water cements are volumetrically minor in the Arab-D in Saudi Arabia.
This zone is a
narrow zone that marks a diffuse boundary between the fresh water
(meteoric) phreatic and marine phreatic diagenetic zones. While Runnels
(1969) has shown that the mixing of two chemically dissimilar
Crystal Growth Contact Inhibition Micropores in microporous cements probably result from the persistence of a thin film of water at crystal interfaces during crystal growth (Wardlaw, 1976). During crystallization, CaCO3 precipitates from interstitial waters; as adjacent crystals grow toward each other, a thin film of water is preserved along crystal faces. Contact between adjacent crystals is actually inhibited by this thin water film, and in this manner, thin “intercrystalline” micropores are developed between cement crystals. Since the vast majority of all diagenetic modifications of carbonate sediments occur in the presence of water (Bathurst, 1975), these thin “intercrystalline” micropores survive subsequent diagenetic modifications of crystal mineralogy. Such “intercrystalline” micropores are observed in the Arab-D and probably occur in most carbonate rocks that contain cement. Although micropores of this type are volumetrically minor, they are significant in that they provide a passage between microporous grains and each other, and to larger macropores. These intercrystalline micropores commonly rim microporous grains, and occur as straight, tubular or laminar pores leading from microporous grains to other microporous grains, or open out into the larger macropore system.
As previously noted, endolithic algae and fungi produce relatively little microporosity in the Arab-D. Although micrite envelopes and borings are locally common, the microporosity formed in these envelopes is insignificant relative to that developed in micritized grains or matrix. Grain alteration and micritization is common in Recent sedimentary environments (Bathurst, 1975). In Recent environments, grain micritization ranges from single micrite-filled borings within grains to complete grain micritization. Although such completely micritized grains superficially appear similar to Arab-D micritized grains, detailed examination of micropore sizes and habit reveals them to be very different (Figures 11d to 11f). Micropores in Arab-D grains typically are straight tubular to laminar pores usually less than 1 micron in diameter; micropores in Recent micritized grains, however, are larger and more equi-dimensional in habit (Figure 11f). Such differences in habit are to be expected, since micropores in Recent sediments occur between micrite crystals of metastable mineralogy and would be extensively altered (if preserved at all) by diagenesis.
Microporosity
occurs almost universally throughout Arab Formation carbonates of Saudi
Arabia. Qualitatively, microporosity occurs in four major categories:
(1) microporous grains, (2) microporous matrix, (3) microporous fibrous
to bladed cements, and (4) microporous equant cements. Quantitative thin
section data reveals that microporosity can vary widely, from 0% to 100%
of the total Capillary
pressure data, while not strictly analogous to the point count data,
also suggest that microporosity comprises a significant portion of the
total We suggest that microporosity in the Arab-D formed via three mechanisms: (1) leaching and incomplete reprecipitation of metastable carbonate (probably caused by fluids derived from the overlying evaporite); (2) crystal growth contact inhibition; and (3) (rarely) endolithic borings of grains.
Appreciation is given to Saudi Aramco and the Ministry of Petroleum and Minerals, Saudi Arabia, for permission to publish this article. We also extend our thanks to Chris Kendall, who reviewed an earlier version of this manuscript, the two anonymous reviewers, and to the GeoArabia staff for their assistance with editing and redesigning the graphics for this paper.
Al-Husseini, M.I. 1997. Jurassic Sequence Stratigraphy of the Western and Southern Arabian Gulf. GeoArabia, v. 2, no. 4, p. 361-380. Bathurst, R.G.C. 1964. The Replacement of Aragonite by Calcite in the Molluscan Shell Wall. In J. Imbrie and N.D. Newell (Eds.), Approaches to Paleoecology. Wiley, New York, p. 357-376. Bathurst, R.G.C. 1975. Carbonate Sediments and their Diagenesis (Developments in Sedimentology 12). Elsevier, Amsterdam-London-New York, 658 p. Bray, R.D. 1997. The Arab-D Reservoir Exposed: Evidence for Vadose Diagenesis in Ghawar Field, Saudi Arabia. Abstract. GeoArabia, v. 2, no. 4, p. 481-482. Bricker, O.P. 1971. Carbonate Cements. John Hopkins Press, Baltimore, 376 p. Broomhall, R.W. and J.R. Allan 1985. Regional Caprock-Destroying Dolomite on the Middle Jurassic to Early Cretaceous Arabian Shelf. Society of Petroleum Engineers, v. 13697, p. 157-160. Budd, D.A. 1989. Micro-Rhombic Calcite and Microporosity in Limestones: A Geochemical Study of the Lower Cretaceous Thamama Group, UAE. Sedimentary Geology, v. 63, p. 293-311. Butler, G.P. 1969. Modern Evaporite Deposition and Geochemistry of Coexisting Brines, the Sabkha, Trucia Coast, Arabian Gulf. Journal of Sedimentary Petrology, v. 39, p. 70-81. Cayeux, L. 1935. Les Roches Sedimentares de France. Roches Carbonatees Masson, Paris, 463 p. Chave, K. 1964. Skeletal Durability and Preservation. In J. Imbrie and N.D. Newell (Eds.), Approaches to Paleoecology. Wiley, New York, p. 377-387. Choquette, P.W. and L.C. Pray 1970. Geological Nomenclature and Classification of Porosity in Sedimentary Cabonates. American Association of Petroleum Geologists Bulletin, v. 54, p. 207-250. Clark, D.N. 1980. The Diagenesis of Zechstein Carbonate Sediments. In H. Fuchtbauer and T. Peryt (Eds.), The Zechstein Basin with Emphasis on Carbonate Sequences. Contributions in Sedimentology 9, p. 167-203. Cloud, P.E. 1962. Environment of Calcium Carbonate Deposition West of Andros Island, Bahamas. United States Geological Survey, Professional Papers, v. 350, p. 1-138. Folk, R.L. 1965. Some Aspects of Recrystallization in Ancient Limestones. In L.C. Pray and R.C. Murray (Eds.), Dolomitization and Limestone Diagenesis: A Symposium. Society of Economic Paleontologists and Mineralogists, Special publication no. 13, p. 14-48. Fyfe, W.S. and J.L. Bischoff 1965. The Calcite-Aragonite Problem. In L.C. Pray and R.C. Murray (Eds.), Dolomitization and Limestone Diagenesis: A Symposium. Society of Economic Paleontologists and Mineralogists, Special Publication no. 13, p. 3-13. Halley, R.B. and P.M. Harris 1979. Fresh Water Cementation of a 1,000 Year-Old Oolite. Journal of Sedimentary Petrology, v. 49, p. 969-988. Hanshaw, B.C., W. Back and R.G. Deike 1971. A Geochemical Hypothesis for Dolomitization by Groundwater. Economic Geology, v. 66, p. 710-724. Harris, P.M. 1979. Facies Anatomy and Diagenesis of a Bahamian Ooid Shoal-Sedimenta VII. University of Miami, Rosenstiel School of Marine and Atmospheric Science, Miami, 163 p. Illing, L.V. 1954. Bahamian Calcareous Sands. American Association of Petroleum Geologists Bulletin, v. 38, p. 1-95. Johnson, J.H. 1971. An Introduction to the Study of Organic Limestones. Quarterly of the Colorado School of Mines, v. 66, no. 2, 185 p. Keith, B.D. and E.D. Pittman 1983. Bimodal Porosity in Oolitic Reservoir Effect on Productivity and Log Response, Rodessa Limestone (Lower Cretaceous), East Texas Basin. American Association of Petroleum Geologists Bulletin, v. 67, p. 1391-1399. Kieke, E.M. and D.J. Hartmann 1974. Detecting Microporosity to Improve Formation Evaluation. Journal of Petroleum Technology, p. 1080-1086. Kobluk, D.R. and M.J. Risk 1977. Micritization and Carbonate-Grain Binding by Endolithic Algae. American Association of Petroleum Geologists, v. 61, p. 1069-1082. Land, L.S. 1973. Contemporaneous Dolomitization of Middle Pleistocene Reefs by Meteoric Water, North Jamaica. Bulletin of Marine Science, v. 23, p. 64-92. Logan, B.W. 1974. Inventory of Diagenesis in Holocene-Recent Carbonate Sediments, Shark Bay, Western Australia. In Logan et al. (Eds.), Evolution and Diagenesis of Quaternary Carbonate Sequences, Shark Bay, Western Australia. American Association of Petroleum Geologists Memoir 22, p. 195-249. Longman, M.W. 1980. Carbonate Diagenetic Textures from Nearsurface Diagenetic Environments. American Association of Petroleum Geologists Bulletin, v. 64, p. 461-487. Loreau, J.-P. and B.H. Pursur 1973. Distribution and Ultrastructure of Holocene Ooids in the Persian Gulf. In B.H. Purser (Ed.), The Shallow Epicontinental Sea. Springer, Berlin, p. 279-328. Lowenstam, H.A. 1963. Biologic Problems Relating to the Composition and Diagenesis of Sediments. In T.W. Donnell (Ed.), The Earth Sciences - Problems and Progress in Current Research. University of Chicago Press, Chicago, p. 137-195. MacClintock, C. 1967. Shell Structure of the Patteloid and Bellerophontoid Gastropods (Mollusca). Peabody Museum, Natural History, Yale University, Bulletin 22, p. 1-140. Majewske, O.P. 1969. Recognition of Invertebrate Fossil Fragments in Rocks and Thin Sections. Brill, Leiden, 101 p. Melim, L.A. and P.K. Swart 1998. Recognizing Marine-Burial Diagenesis: Criteria and Examples from Quaternary to Neogene Platform to Slope Carbonates, Bahamas and Florida. Presented at the 1998 Annual American Association of Petroleum Geologists Meeting. American Association of Petroleum Geologists Annual Meeting (Program with Abstracts). Meyer, F.O. and R.C. Price 1992. A New Arab-D Depositional Model, Ghawar Field, Saudi Arabia. Society of Petroleum Engineers Paper No. 25576, p. 1-10. Meyers, J. and K.C. Lohmann 1978. Microdolomite-Rich Syntaxial Cements: Proposed Meteoric – Marine Mixing Zone Phreatic Cements from Mississippian Limestones, New Mexico. Journal of Sedimentary Petrology, v. 48, p. 475-488. McGill, M.M. and K.R. Walker 1982. Diagenetic Study of the Holston Limestone: Paragenesis of Cements and Evaluation of Geochemistry. Presented at 1982 Annual Meeting Geological Society of America Southeast and Northeast Sections. Geological Society of America Southeast and Northeast Annual Meeting (Program with Abstracts). Mitchell, J.C., P.J. Lehmann, D.L. Cantrell, I.A. Al-Jallal and M.A.R. Al-Thagafy 1988. Lithofacies, Diagenesis and Depositional Sequence; Arab-D Member, Ghawar Field, Saudi Arabia. In A.J. Lomando and P.M. Harris (Eds.), Giant Oil and Gas Fields - A Core Workshop. Society of Economic Paleontologists and Mineralogists, Core Workshop no. 12, p. 459-514. Moore, C.M. and Y. Druckman 1981. Burial Diagenesis and Porosity Evolution, Upper Jurassic Smackover, Arkansas and Louisiana. American Association of Petroleum Geologists Bulletin, v. 65, p. 597-628. Moshier, S.O. 1989. Development of Microporosity in a Micritic Limestone Reservoir, Lower Cretaceous, Middle East. Sedimentary Geology, v. 63, p. 217-240. Newell, N.D., E.G. Purdy and J. Imbrie 1960. Bahamian Oolitic Sand. Journal of Geology, v. 68, p. 481-497. Newell, N.D. and J.K. Rigby 1957. Geological Studies on the Great Bahama Bank. In R.J. LeBlanc and J.G. Breeding (Eds.), Regional Aspects of Carbonate Deposition. Society of Economic Paleontologists and Mineralogists, Special Publication no. 5, p. 15-72. Oldershaw, A.E. 1972. Microporosity Control in Microcrystalline Carbonate Rocks, Southern Ontario, Canada. In Stratigraphy and Sedimentology, International Geological Congress Proceedings, Programme No. 24, p. 198-207. Palmer, T.J., J.D. Hudson and M.A. Wilson 1988. Palaeoecological Evidence for Early Aragonite Dissolution in Ancient Calcite Seas. Nature, v. 335, p. 809-810. Pittman, E.D. 1971. Microporosity in Carbonate Rocks. American Association of Petroleum Geologists Bulletin, v. 55, p. 1873-1881. Purser, B.H. 1969. Syn-Sedimentary Marine Lithification of Middle Jurassic Limestones in the Paris Basin. Sedimentology, v. 12, p. 205-230. Runnels, D.D. 1969. Diagenesis, Chemical Sediments, and the Mixing of Natural Waters. Journal of Sedimentary Petrology, v. 39, p. 1188-1201. Sandberg, P.A. 1975. New Interpretation of Great Salt Lake Ooids and of Ancient Non-Skeletal Carbonate Mineralogy. Sedimentology, v. 25, p. 673-702. Sandberg, P.A. 1983. An Oscillating Trend in Phanerozoic Non-Skeletal Carbonate Mineralogy. Nature, v. 305, p. 19-22. Sarg, J.R. 1982. Off-Reef Salina Deposition (Silurian), Southern Michigan Basin, Implications for Reef Genesis. In Handford et al. (Eds.), Depositional and Diagenetic Spectra of Evaporites - A Core Workshop. Society of Economic Paleontologists and Mineralogists, Core Workshop no. 3, p. 354-383. Schmidt, V. and D.A. McDonald 1979. The Role of Secondary Porosity in the Course of Sandstone Diagenesis. Society of Economic Paleontologists and Mineralogists, Special Publication no. 26, p. 175-208. Schreiber, B.C., D.J. Kinsman and R.D. Nurmi 1976. Development of Porosity in Carbonate Rocks of Evaporitic Basins, Alternative Mode of Formation. American Association of Petroleum Geologists Bulletin, v. 60, p. 720. Shearman, D.J. 1978. Evaporites of Coastal Sabkhas. In W.E. Dean and B.C. Schreiber (Eds.), Marine Evaporites. Society of Economic Paleontologists and Mineralogists, Short Course No. 4, p. 6-42. Stockman, K.W., R.N. Ginsburg and E.A. Shinn 1967. The Production of Lime Mud by Algae in South Florida. Journal of Sedimentary Petrology, v. 37, p. 633-648. Taft, W.H., F. Arrington, A. Haimovitz, C. MacDonald and C. Woolheater 1968. Lithification of Modern Carbonate Sediments at Yellow Bank, Bahamas. Bulletin of Marine Science Gulf Caribbean, v. 18, p. 762-828. Tissot, B., B. Dunard, J. Espitalic and A. Combay 1974. Influences of Nature and Diagenesis of Organic Matter in Formation of Petroleum. American Association of Petroleum Geologists Bulletin, v. 58, p. 499-506. Wardlaw, N.C.
1976. Wigley, T.M.L. and L.N. Plummer 1976. Mixing of Carbonate Waters. Geochimien et Cosmochimica Acta, v. 40, p. 989-995. Wilkinson, B.H., R.M. Owen and A.R. Carroll 1985. Submarine Hydrothermal Weathering, Global Eustasy, and Carbonate Polymorphism in Phanerozoic Marine Oolites. Journal of Sedimentary Petrology, v. 55, p. 171-183. Wilson, A.O. 1985. Depositional and Diagenetic Facies in the Jurassic Arab-C and -D Reservoirs, Qatif Field, Saudi Arabia. In P.O. Roehl and P.W. Choquette (Eds.), Carbonate Petroleum Reservoirs: A Casebook. Springer-Verlag, New York, p. 319-340. Wilson, J.L. 1975. Carbonate Facies in Geologic History. Springer-Verlag, New York, 471 p. Wray, J.L. 1977. Calcareous Algae (Developments in Paleontology and Stratigraphy No. 4). Elsevier, New York, 185 p.
Complete Captions for Figures 3,4,5,6,7, & 11) Figure 3 a,b,c.d. Microporosity in Arab Formation carbonates. In thin section, porosity is filled by blue-dyed epoxy, calcite is stained pink to red, other minerals are unstained. (a)
Thin
section photomicrograph of skeletal-oolitic grainstone. Interparticle
(BP) and moldic (MO) macropores are abundant. Microporosity (MC) occurs
within micritized grains and foraminifera (F). Isopachous fringing
calcite cement (arrows) coats most grains in this sample
(plane-polarized light, Saudi Arabia, Abqaiq field, Arab-D). (b) SEM
photograph of epoxy Figure 3 e,f.
(e) Epoxy Figure 4 a,b. Microporous grains in Arab Formation carbonates. In thin section, porosity is filled by bluedyed epoxy, calcite is stained pink to red, other minerals are unstained. (a) Thin section photomicrograph of microporous peloids (P) and microporous miliolid foraminifera (M). Microporosity appears as a blue haze within grains (plane-polarized light, unspecified Mesozoic reservoir, Middle East). (b) Thin section photomicrograph of microporous ooid. Microporosity in this grain is denoted by a blue hazy appearance resulting from infiltration of blue-dyed epoxy into micropores in the grain (plane-polarized light, Saudi Arabia, Qatif field, Arab-C). Figure 4 c,d. (c) SEM image of fractured rock chip showing internal fabric of a microporous foraminifer (F). Foraminifer consists of subhedral crystals of micrite ranging from 1 to 10 microns in size. Foraminifer is recognizable by a distinctive chambered pattern of intraparticle porosity (arrows) (Saudi Arabia, Berri field, Arab-C). (d) Higher magnification view of foraminifer shown in (c) (SEM image of fractured rock chip). Figure 4 e,f.
(e) SEM image of epoxy Figure 5 a,b. Microporous
grains and microporous matrix in Arab Formation carbonates. In thin
section, porosity is filled by blue-dyed epoxy, calcite is stained pink
to red, other minerals are unstained. (a) Epoxy Figure 5
c,d,e,f. (c)
Thin
section photomicrograph of microporous composite grain (plane-polarized
light, Saudi Arabia, Qatif field, Arab-C). (d) Epoxy Figure 6.
Microporous matrix and microporous cements in Arab Formation
carbonates. Porosity in thin section is filled by blue-dyed epoxy,
calcite is stained red to pink, other minerals are unstained. (a) SEM
view of fractured rock surface of chalky microporous matrix. Microporous
matrix is composed of well-sorted calcite crystals generally 1 to 4
microns in size; microporosity occurs as small pores between these
micrite crystals (unspecified Mesozoic reservoir, Middle East). (b) Thin
section photomicrograph of packstone with abundant moldic (MO) porosity
and common microporous matrix (arrows). Moldic porosity is the result of
leaching of the dasycladacean algae Salpingoporella (DS) which
forms characteristic circular pores. Minor interparticle porosity is
present (plane-polarized light, Saudi Arabia, Berri field, Arab-C). (c)
SEM image of epoxy Figure 7 a,b.
Microporous cements in Arab Formation carbonates. Porosity in thin
section is filled by blue-dyed epoxy, calcite is stained pink to red,
other minerals are unstained. (a) SEM image of epoxy Figure 7
c,d,e,f. (c) Thin
section photomicrograph of grainstone with abundant equant calcite
cement (arrows). This cement fills interparticle porosity between
microporous micritized grains (MG) and probable composite grain (CG)
(plane-polarized light, Saudi Arabia, Berri field, Arab-C). (d) Epoxy
Figure 11 a,b,c,d. Microporous grains in the Jurassic and in the Recent. Porosity in thin section is filled by blue-dyed epoxy, calcite is stained pink to red, other minerals are unstained. (a) Thin section photomicrograph of ooid-skeletal mud-lean packstone. Large brachiopod fragment (B) was originally composed of low magnesium calcite and is non-microporous. In contrast, surrounding grains are micritized and microporous (plane-polarized light, Saudi Arabia, Khursaniyah field, Arab-D). (b) Thin section photomicrograph showing micritized and microporous grains originally composed of metastable high magnesium calcite. Original high magnesium calcite components include several types of foraminifera (F), micritized grains (MG) and the red algae Thaumatoporella (TP) (plane-polarized light, Saudi Arabia, Ghawar field, Arab-D). (c) Thin section photomicrograph showing preserved microstructure of several echinoderm grains (E). In spite of their original high magnesium calcite mineralogy, echinoderms are rarely microporous. Algal or fungal borings (B) and micrite envelopes (arrows) are locally common on these grains, although these grains never appear completely micritized (plane-polarized light, Saudi Arabia, Ghawar field, Arab-D). (d) Photomicrograph of a Recent skeletalpeloidal sand. Peloids (P) are completely micritized and microporous, as indicated by their bluish color. Blue appearance results from the infiltration of blue-dyed epoxy into micropores in the grains. In contrast, solid grains are pearly white in color. Note that some grains are incompletely micritized (arrows), and grade from bluish micritized and microporous CaCO3 into white, solid CaCO3 within the same grain (reflected light, Holocene, North Caicos, British West Indies). Figure 11 e,f.
(e) SEM photograph of epoxy
ABOUT THE AUTHORS
|

 Figure 1a
Figure 1a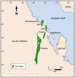 Figure 1b
Figure 1b Figure 2
Figure 2 **Figure 3
a,b,c,d.
**Figure 3
a,b,c,d.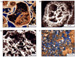 **Figure 5
c,d,e,f
**Figure 5
c,d,e,f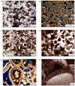 **Figure 6
**Figure 6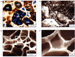 **Figure 7
c,d,e,f
**Figure 7
c,d,e,f Figure 8
Figure 8 Figure 10
Figure 10
 Figure 12
Figure 12 Figure 13
Figure 13 Table 1
Table 1