Click to view article in PDF format.
Click to view article in PDF format.
GC Energy
Energy Density of Deepwater Gas Hydrate*
Density of Deepwater Gas Hydrate*
By
Bob Hardage1
Search and Discovery Article # 40241 (2007)
Posted May 23, 2007
*Adapted from the Geophysical Corner column, prepared by the author, in AAPG Explorer, May, 2007, and entitled “Gas Hydrate and LNG Tankers”. Editor of Geophysical Corner is Bob A. Hardage. Managing Editor of AAPG Explorer is Vern Stefanic; Larry Nation is Communications Director.
1Bureau of Economic Geology, The University of Texas at Austin ([email protected] )
General Statement
Theoretical models have been developed at the Bureau of Economic Geology that relate formation velocity and resistivity to hydrate concentration (Cgh) in deepwater, near-seafloor sediments. Our studies indicate that in numerous targeted intervals across the Gulf of Mexico, Cgh is 0.5 to 0.6 of the available pore space in unconsolidated deepwater sediments.
The reaction of most explorationists to this finding is “Too bad. That gas concentration is too low to be of interest.” This conclusion is logical for anyone whose experience has been only with conventional gas reservoirs, where gas concentrations of 50 to 60 percent are not appealing. It may not be a correct conclusion for gas hydrate reservoirs.
|
uHydrate
vs LNG
uHydrate
vs LNG
uHydrate
vs LNG
uHydrate
vs LNG
uHydrate
vs LNG
|
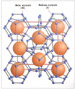
Formation of HydrateLet’s consider how the formation of hydrate causes a high concentration of natural gases by comparing the physical sizes of a sediment grain size and a unit-volume of hydrate. A unit-volume of Structure I hydrate is shown as Figure 1. Limited page space does not permit the unit-volume geometries of Structure II and Structure H hydrates to be illustrated. This crystalline structure is called a “unit-volume” because Structure I hydrate grows in increments of this fundamental building block. This unit-volume consists of eight cages of structured water that can each trap one gas molecule. Dendy Sloan at the Colorado School of Mines (e.g., Sloan, 2003, 2004) defines the diameter of each cage of this unit-volume as a length that varies from 8 to 10 angstroms. Because any arbitrary diameter across this unit-volume will span no more than six cages (probably no more than four cages, actually), the diameter of a unit-volume of hydrate is less than 60 angstroms (6 x 10-9 m). For a size comparison, sedimentologists define the low end of very fine-grained sand to be fragments of sediment that have diameters of about 60 microns (micrometers) (6 x 10-5 m). The diameter of very fine-grained sand is thus larger than the diameter of a unit-volume of hydrate by a factor of 104. Because the volume of an object is proportional to (diameter)3, if we ratio the volume of a very fine-grained sand and the volume of a unit-volume of hydrate, we find that this 104 difference in diameters means that 1012 unit-volumes of hydrate can fit into the space occupied by one very fine sand grain. Assuming that on average only 80 percent of the eight cages in a unit-volume manage to trap a gas molecule, hydrate formation causes approximately 6 x 1012 gas molecules to be compressed into a volume equal to that of a single very fine sand grain.
Gas Hydrate vs LNG
|