Click to view article as PDF
Click to view article as PDF
Overview of Porosity Evolution in Carbonate Reservoirs*
By
S. J. Mazzullo1
Search and Discovery Article #40134 (2004)
*This online version is basically a reprint of an article by the author and with the same title, published in Kansas Geological Society Bulletin, v. 79, nos. 1 and 2 (January-February and March-April, 2004) and available on the KGS website (http://www.kgslibrary.com/bulletins/bulletins.htm). Appreciation is expressed to the author and to KGS for permission to present this article on Search and Discovery.
1Department of Geology Wichita State University, Wichita, Kansas ([email protected])
Introduction
Carbonate rocks (limestone and
dolomite) account for approximately 50% of oil and gas production around the
world. Of the carbonates, a slightly greater percentage of world  hydrocarbon
hydrocarbon reserves has been produced from dolomites because such rocks commonly, but not
always, have more porosity and permeability than limestones (Halley and Schmoker,
1983). Unlike most sandstone reservoirs, which typically are single-porosity
systems (i.e., interparticle pores) of relative uniform (homogeneous) nature,
reservoirs in carbonate rocks commonly are multiple-porosity systems that
characteristically impart petrophysical heterogeneity to the reservoirs (Mazzullo
and Chilingarian, 1992). Hence, the specific types and relative percentages of
pores present, and their distribution within the rocks, exert strong control on
the production and stimulation characteristics of carbonate reservoirs (e.g.,
Jodry, 1992; Chilingarian et al., 1992; Honarpour et al., 1992; Hendrickson et
al., 1992; Wardlaw, 1996). Pore types in carbonate rocks can generally be
classified on the basis of the timing of porosity evolution (Choquette
and Pray, 1970) into: (1) primary pores (or depositional porosity), which are
pores inherent in newly-deposited sediments and the particles that
comprise them. Such pore types include interparticle pores in, for
example, carbonate sands (but also in muddy carbonates), intraparticle
pores (within particles such as foraminifera or gastropod shells), fenestral
pores (formed by gas bubbles and sediment shrinkage in tidal-flat carbonates),
and shelter and growth-framework pores (common in reef buildups);
and (2) secondary pores, which are those that form as a result of later,
generally post-depositional dissolution. Such pore types include all of those
mentioned above, but only when it can be demonstrated that primary pores which
subsequently were occluded by cement later had all or some of that cement
dissolved (resulting in the generation of exhumed pores - Figure 2), as well as
vugs (large pores that transect rock fabric, that is, dissolution was not
fabric-selective) and dissolution-enlarged fractures. Most of these
primary and secondary pore types can readily be identified in cores, and with
the possible exception of shelter and growth-framework pores, also in well
cuttings samples.
reserves has been produced from dolomites because such rocks commonly, but not
always, have more porosity and permeability than limestones (Halley and Schmoker,
1983). Unlike most sandstone reservoirs, which typically are single-porosity
systems (i.e., interparticle pores) of relative uniform (homogeneous) nature,
reservoirs in carbonate rocks commonly are multiple-porosity systems that
characteristically impart petrophysical heterogeneity to the reservoirs (Mazzullo
and Chilingarian, 1992). Hence, the specific types and relative percentages of
pores present, and their distribution within the rocks, exert strong control on
the production and stimulation characteristics of carbonate reservoirs (e.g.,
Jodry, 1992; Chilingarian et al., 1992; Honarpour et al., 1992; Hendrickson et
al., 1992; Wardlaw, 1996). Pore types in carbonate rocks can generally be
classified on the basis of the timing of porosity evolution (Choquette
and Pray, 1970) into: (1) primary pores (or depositional porosity), which are
pores inherent in newly-deposited sediments and the particles that
comprise them. Such pore types include interparticle pores in, for
example, carbonate sands (but also in muddy carbonates), intraparticle
pores (within particles such as foraminifera or gastropod shells), fenestral
pores (formed by gas bubbles and sediment shrinkage in tidal-flat carbonates),
and shelter and growth-framework pores (common in reef buildups);
and (2) secondary pores, which are those that form as a result of later,
generally post-depositional dissolution. Such pore types include all of those
mentioned above, but only when it can be demonstrated that primary pores which
subsequently were occluded by cement later had all or some of that cement
dissolved (resulting in the generation of exhumed pores - Figure 2), as well as
vugs (large pores that transect rock fabric, that is, dissolution was not
fabric-selective) and dissolution-enlarged fractures. Most of these
primary and secondary pore types can readily be identified in cores, and with
the possible exception of shelter and growth-framework pores, also in well
cuttings samples.
Because the natural tendency in most
carbonate sediments is that primary porosity is substantially reduced by
cementation and compaction during post-depositional burial (Figure 1; Halley and
Schmoker, 1983), many workers would argue that most porosity in limestone and
dolomite reservoirs is of secondary origin (e.g., Mazzullo and Chilingarian,
1992). Exceptions to this statement are cases where primary porosity is
preserved because of the early influx of hydrocarbons into pores (e.g., Feazel
and Schatzinger, 1985). Early on in the study of carbonate sediments and their
diagenesis, the subaerial meteoric diagenetic (freshwater) model was
promoted as a means of explaining porosity evolution in carbonates, specifically
in shallow-water carbonates that lie beneath unconformities in paleo-vadose and
paleo-phreatic freshwater zones (e.g., Friedman, 1964; Land, 1967). This model
still is heavily applied today, especially in sequence stratigraphic-related
diagenetic studies of reservoirs. This model presupposes that if porous
carbonate rocks are present beneath unconformities, then that porosity must have
been created by freshwater dissolution during subaerial exposure. Of course, as
explorationists, we all can probably list a large number of wells that were
drilled into non-porous carbonate rocks beneath unconformities. Hence, the
corollary pertaining to subaerial exposure is not true - meteoric exposure does
not always create porosity, and even if it did, that porosity may be occluded
during later burial (Figure 1). A most critical constraint on evaluating, or
more importantly, on  predicting
predicting porosity in carbonate rocks utilizing only the subaerial meteoric diagenetic model is that one must call upon fluids capable of
dissolving carbonate to come from above; that is, from rain water
percolating down into sediments or rocks beneath unconformity surfaces.
Ostensibly, then, many might consider that if there is not an unconformity in
the section, then the carbonates will not be porous. Again, as explorationists
we can all probably compile a list of wells in which porous carbonates that were
not associated with unconformities were encountered in the subsurface.
porosity in carbonate rocks utilizing only the subaerial meteoric diagenetic model is that one must call upon fluids capable of
dissolving carbonate to come from above; that is, from rain water
percolating down into sediments or rocks beneath unconformity surfaces.
Ostensibly, then, many might consider that if there is not an unconformity in
the section, then the carbonates will not be porous. Again, as explorationists
we can all probably compile a list of wells in which porous carbonates that were
not associated with unconformities were encountered in the subsurface.
The foregoing analysis therefore begs
the following questions: (1) can secondary porosity in carbonate rocks be
generated by processes other than subaerial meteoric exposure, and if so,
what are those processes?; (2) how might reservoir porosity formed by such
alternative processes differ from reservoirs created by meteoric dissolution
along unconformities?; (3) how can we recognize and determine the origin of
reservoir porosity?; and (4) can the subsurface occurrence of porosity formed by
any such alternative models of reservoir origin be predicted? The purpose of
this contribution is to address these questions by demonstrating the
multi-faceted evolution of secondary porosity in carbonate  hydrocarbon
hydrocarbon reservoirs. In the following discussions attention will be focused on the
recognition and origin of pore types in shallow-water limestones and dolomites,
as observed mainly in cores and well cuttings samples.
reservoirs. In the following discussions attention will be focused on the
recognition and origin of pore types in shallow-water limestones and dolomites,
as observed mainly in cores and well cuttings samples.
|
|
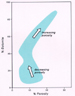
Secondary Porosity Beneath Unconformities: The Subaerial Meteoric Model The generation of secondary porosity in carbonate sediments or rocks in this model is a direct consequence of dissolution by freshwater (ultimately rain-derived), which dissolves carbonates because the water is undersaturated with respect to calcium carbonate. The extent of dissolution and secondary porosity formation are controlled by factors such as the acidity of freshwater (e.g., rain water percolating down through a soil zone will be more acidic than in areas where soils are not present), the amount of porosity or fractures within the affected carbonates, the residence time of the freshwater in the diagenetic system, the mineralogy of the sediments or rocks, and so forth (Longman, 1980; Moore, 1989). Secondary porosity generation via dissolution can occur relatively soon after deposition, in unconsolidated sediments, in what Choquette and Pray (1970) refer to as the eogenetic zone; or it can occur much later, in rocks, in the telogenetic zone as a consequence of uplift of older, formerly buried carbonates (Figure 2). In newly-deposited carbonate sediments that subsequently are subaerially exposed, it is the difference in original mineralogies of particles in the sediments that drives the relatively rapid, selective dissolution of particles. Fragments of corals, pelecypods and gastropods, and oolites, for example, are originally composed mostly of the mineral aragonite (CaCO3, orthorhombic), which is very soluble. It is for this reason that formerly aragonitic particles in limestones usually are represented by pores (in this case, fabric-selective pores) or cement-filled pores. In contrast, particles such as forams, crinoid fragments, and bryozoans are originally composed of the mineral high-magnesium calcite (CaCO3, hexagonal-rhombohedral), which is calcite with up to 23 mole% MgCO3 in the crystal lattice. With exposure to freshwater such particles tend merely to lose MgCO3 and not to dissolve like aragonitic particles. Other particles, such as brachiopod shells and some pelecypods, build their skeletons out of low-Mg calcite (also CaCO3, hexagonal-rhombohedral), which is calcite with less than 4 mole% MgCO3. Particles of original high-magnesium calcite and low-magnesium calcite mineralogy tend not to dissolve unless the freshwater is quite undersaturated with respect to calcium carbonate, and it is for this reason that some particles (crinoids, bryozoans, brachiopods) often are well-preserved in ancient rocks. The eogenetic exposure to freshwater of newly-deposited carbonate sediments, which are generally highly porous and polyminerallic (i.e., as discussed above, composed of mixtures of aragonite, high-magnesium and low-magnesium calcite), results in the formation of cemented limestones, of varying porosity, of stable low-magnesium calcite composition (notwithstanding dolomitization). In contrast, late eogenetic or telogenetic freshwater exposure of older limestones that have already been mineralogically stabilized and cemented is not driven by such differences in the relative solubility of aragonite, high-magnesium and low-magnesium calcite because the rocks already are mineralogically stabilized to low-magnesium calcite, and further dissolution can occur only if the fluids are quite undersaturated with respect to calcite (the least soluble of the aforementioned carbonate minerals). Typically, such dissolution forms vugs and caverns, which can also form in polyminerallic carbonate sediments. Dissolution of already stabilized limestones can also result in the formation of particle-selective pores when certain particles in the rocks are slightly more soluble than other particles because of differences in particle size or their micro-architectural arrangement of component calcite crystals. For example, crinoid fragments in older rocks are composed of single, relatively large crystals of low-magnesium calcite, which have relatively low solubility. It is for this reason that crinoid-rich Mississippian limestones, for example, typically have low porosities. In contrast, foram shells are composed of myriads of small calcite crystals, which have relatively higher solubility, and they usually are more readily dissolved than crinoid fragments. In either case, it is important to note that carbonates can be affected by meteoric dissolution not only directly beneath unconformities on land, but also for some distance down-dip into the subsurface ("A" in Figure 2) and some distance in a seaward direction below sea level, depending on the extent of freshwater lenses ("B" in Figure 2). Porosity generation by dissolution eventually ceases, generally in a down-dip direction within phreatic zones when that water becomes saturated with respect to dissolved calcium carbonate. At that point, porosity can be maintained, or if the water becomes even more saturated with respect to dissolved calcium carbonate, it can begin to be occluded by carbonate cement (and other cements as well, such as gypsum/anhydrite or silica).
Meteoric Porosity in Limestones
In
limestones, common secondary pore types formed as a result of
post-depositional dissolution variously include exhumed interparticle,
intraparticle, fenestral, shelter, and growth-framework pores, all of
which are considered to be fabric-selective pores; and also not
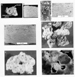
fabric-selective vugs (Figure 3E) and dissolution-enlarged fractures.
The size of vugs (Figure 4) varies from small (but larger than component
particles in the rocks) to caverns or cavernous porosity. Vugs may
originate either by wholescale dissolution of parts of the rock or by
dissolutional enlargement of fabric-selective pores (Figure 3E). In many
cases there is coincidence between the types of fabric-selective pores
present in the rocks and the depositional environment of the rocks,
which serves as an important guide in evaluating permeability and
potential recoverable reserves from the reservoir, and in deciding on
what stimulation procedures to use but only if one knows the
depositional environment of the rocks from study of subsurface samples.
For example, carbonate sands (lime grainstones), deposited in
high-energy environments such as oolite shoals or skeletal sand shoals,
commonly have high interparticle porosity (Figure 3A) and attending
relatively high permeabilities. On the other hand, however, high
porosity but low permeability may characterize a carbonate sand
(limestone) reservoir wherein only the particles have been dissolved
(for example, in cases where the reservoir contains only molds of
oolites referred to as oomoldic porosity or by the older term oocastic
porosity: Figure 3D). In such cases there may be ample Over-riding such generalizations about the relationships among pore types, permeability, and depositional environments of the limestones is the importance of pore throats in the rocks (Wardlaw, 1976). In limestones, particularly in grainstones, for example, the nature of pore throats and their effect on permeability is controlled by the size of the particles in the rocks, and more importantly, by the distribution of any remaining earlier-precipitated cement in the pores that was not dissolved (Figure 3F). Calcite cement overgrowths on crinoid fragments can significantly restrict pore throats as well (Figure 5), which is why many crinoid-rich Mississippian limestones are of low-permeability nature. The best way to determine the extent of pore-throat restriction in the rocks under consideration is by examining the rocks petrographically in thin-section. Clay-mineral cements are extremely rare in carbonate reservoir rocks, and therefore, need not be considered here.
Meteoric Porosity in Dolomites In contrast to earlier postulates on the subject (e.g., Murray, 1960; Weyl, 1960), the process of dolomitization of a pre-existing limestone does not automatically create secondary porosity. Whereas it is true that porosity tends to increase as amount of dolomite increases (Figure 6), it generally does so for the following reasons. In partly dolomitized limestones exposed to telogenetic meteoric fluids, for example, any remaining calcite (which may represent particles and/or carbonate mud matrix) inherently is more susceptible to dissolution by freshwater because it is more soluble than dolomite. Hence, subaerial exposure of a partly dolomitized limestone can result in the generation of the same types of pores as described above by dissolution of remaining calcite, depending on the original texture of the rock (mudstone, wackestone, packstone, or grainstone), its depositional environment, and degree of replacement by dolomite (Figure 7). Likewise, remaining evaporites in the rocks can also be dissolved. In more pervasively dolomitized rock exposed to telogenetic meteoric fluids, remaining calcite (or evaporite minerals) between dolomite crystals can be dissolved during subaerial exposure to produce intercrystalline pores between dolomite crystals. In completely dolomitized rocks, vugs (and sometimes dissolution- enlarged fractures) are common pore types present if the meteoric fluids were highly acidic or acted on the rocks over long periods of time. Selective dissolution of small dolomite crystals (because solubility increases as crystal size decreases), or of more soluble dolomite phases in the rocks, can result in the development of vugs and intercrystalline pores. All such processes and resulting pore types can be represented in a given reservoir. As in limestones, the nature of pore throats in dolomites affects permeability, and as a general rule, intercrystalline pore throat sizes decrease with decreasing crystal size and extent of dolomitization (Figure 8).
Cavernous Porosity in Carbonate Rocks
Cavernous
and associated vuggy porosity are major attributes of
Alternative Origin Of Secondary Porosity: The Mesogenetic Model
Since the
late 1970s-early 1980s, geologists began to suspect that not all
secondary dissolution porosity in carbonate rocks forms or formed solely
beneath unconformities by freshwater dissolution in either the eogenetic
or telogenetic environments (e.g., Bathurst, 1980; Scholle and Halley,
1985; Choquette and James, 1987; Moore, 1989). Rather, there was growing
realization of the significance of, and processes controlling, secondary
dissolution porosity formation (and porosity occlusion) in the
deep-burial environment B which is what Choquette and Pray (1970)
referred to as the mesogenetic environment. Two important points
in this regard are the facts that: (1) not all porous carbonates are
associated with unconformities; and (2) specifically, there are a number
of examples of porous and permeable carbonate rocks deposited in
deep-water settings and which later were deeply buried and never
subaerially exposed. Hence, meteoric exposure at any time after
deposition has been ruled out for such rocks (e.g., Mazzullo and Harris,
1991; Mazzullo, 1994). Therefore, post-depositional diagenesis and the
formation of secondary dissolution porosity in such rocks must have
occurred in an environment other than the meteoric eogenetic or
telogenetic environment. Furthermore, if we are exploring for
As discussed above, in order for dissolution of any carbonate rocks to proceed, they must be exposed to fluids that are undersaturated with respect to calcium carbonate. That is easy enough to do in the subunconformity meteoric environment because rain water, the ultimate source of near-surface freshwater, is undersaturated. In the mesogenetic environment, however, most connate fluids are brines that typically are saturated or even supersaturated with respect to calcium carbonate, which means they are not capable of dissolving carbonate rocks and creating secondary porosity. Rather, such fluids tend to precipitate carbonates in the form of calcite or dolomite cement, and in some cases, they may be capable of dolomitization. How, then, can carbonate dissolution and porosity formation proceed in the deep-burial environment? In other words, how are fluids undersaturated with respect to calcium carbonate generated in the mesogenetic environment?
Studies of
porosity evolution in sandstones, combined with studies of organic
matter maturation and
Porosity
formed in the mesogenetic environment is represented by the same types
of pores that can form in the eogenetic and telogenetic freshwater
environment (Mazzullo and Harris, 1992), including even cavernous
porosity (e.g., Hall, 1990; Hill, 1992), which otherwise is generally
known as A burial karst. Therefore, an important point to
remember in this regard is that the diagenetic environment in which
porosity formation occurred cannot be determined on the basis of the
pore types present in a reservoir! How, then, does one recognize
porosity formed in the mesogenetic environment? The answer to this
question is "With careful, detailed petrographic study of
thin-sections, often combined with analysis of carbon and oxygen
isotopic values of carbonate cements associated with suspected
mesogenetic porosity.” For details, refer to the papers by Moore and
Druckman (1981), Druckman and Moore (1985), Heydari and Moore (1989),
Moore (1989) and Mazzullo and Harris (1991, 1992), and the many papers
cited therein. There are, however, some readily visible clues to the
mesogenetic origin of porosity in carbonate rocks that can be identified
in cuttings and core samples. For example: (1) porosity along and
associated with stylolites or which cuts across stylolites (Figure 13).
As pressure-dissolution features, stylolites form most commonly in the
deep-burial environment. So, if the timing of porosity formation can be
determined by the law of cross-cutting relationships to be younger
than the stylolites, and the rocks are still buried, then porosity
formation must be the result of mesogenetic dissolution; (2) pores that
cut across cements that contain
Microporosity, Pinpoint Porosity, and Chalky Porosity The term microporosity refers to any very small pores that can be recognized only with the aid of a high-powered binocular microscope or thin-section (Choquette and Pray, 1970; Pittman, 1971). Micropores, otherwise known as pinpoint pores, may variously represent: (1) birdseye pores in tidal flat deposits; (2) intraparticle pores within small particles; (3) intercrystalline pores between dolomite crystals or between calcite cement crystals; (4) intercrystalline pores within the nuclei or cortices of oolites; or (5) intracrystalline pores within individual dolomite or calcite cement crystals. In some cases, whereas matrix microporosity/pinpoint porosity may not be very permeable for oil, it very well may be permeable enough for natural gas (e.g., Roehl, 1985; Ruzyla and Friedman, 1985). Chalky porosity is a term that refers to microporosity that commonly forms in highly weathered or otherwise highly diagenetically altered carbonate rocks (Pittman, 1970; Reeckmann and Friedman, 1982) that, as a consequence of being strongly altered, are very soft. Although true chalks (i.e., those limestones of Cretaceous to Paleogene age that are composed dominantly of coccoliths) commonly contain microporosity, the presence of chalky porosity does not necessarily indicate that the rocks under consideration are chalks. In fact, tripolitic cherts commonly have microporosity, and because the rocks are relatively soft, such porosity can be referred to as chalky. I point out that microporosity, and specifically chalky porosity, can form in carbonate rocks (and cherts) beneath unconformities as well as in deeply buried rocks, and as such, their presence does not necessarily imply the existence of a stratigraphically-nearby unconformity.
ConclusionsSecondary porosity in carbonate rocks, in both limestone and dolomite, can be formed by: (1) freshwater dissolution either in the subunconformity meteoric, eogenetic or telogenetic environment, or; (2) by dissolution by chemically aggressive subsurface fluids, generated during maturation of organic matter in source rocks, in the deep-burial (mesogenetic) environment. Although pore types formed in these environments are similar, their origin often can be determined by careful observation, thin-section petrography, and stable carbon-oxygen isotope analysis.
Selected ReferencesBathurst, R.G.C., 1980, Deep crustal diagenesis in limestones: Revista Instituto Investaciones Geologicas, University of Barcelona, v. 34, p. 89-100.
Burruss, R.C., Cercone, K.R., and
Harris, P.M., 1985, Timing of Cathles, L.M., and Smith, A.T., 1983, Thermal constraints on the formation of Mississippi Valley-type lead-zinc deposits and their implications for episodic basin dewatering and deposit genesis: Economic Geology, v. 78, p. 983-1002. Chilingarian, G.V., Torabzadeh, J., Rieke, H.H., Metghalchi, M., and Mazzullo, S.J., 1992, Interrelationships among surface area, permeability, porosity, pore size, and residual water saturation, in G.V. Chilingarian, S.J. Mazzullo, and H.H. Rieke, eds., Carbonate Reservoir Characterization: A Geologic-Engineering Analysis, Part I: Elsevier Publ. Co., Amsterdam, Developments in Petroleum Science 30, p. 379-397. Choquette, P.W., and James, N.P., 1987, Diagenesis 12: diagenesis in limestones-3. The deep burial environment: Geoscience Canada, v. 14, p. 3-35. Choquette, P.W., and Pray, L.C., 1970, Geologic nomenclature and classification of porosity in sedimentary carbonates: AAPG Bulletin, v. 54, p. 207-250. Clendenin, C.W., and Duane, M.J., 1990, Focused fluid flow and Ozark Mississippi Valley-type deposits: Geology, v. 18, p. 116-119. Druckman, Y., and Moore, C.H., 1985, Late subsurface porosity in a Jurassic grainstone reservoir, Smackover Formation, Mt. Vernon field, southern Arkansas, in P.O. Roehl and P.W. Choquette, eds., Carbonate Petroleum Reservoirs: Springer-Verlag, New York, p. 371-383. Feazel, C.T., and Schatzinger, R.A., 1985, Prevention of carbonate cementation in petroleum reservoirs, in N. Schneidermann and P.M. Harris, eds., Carbonate Cements: SEPM Special Publ. 36, p. 97-106. Foscolos, A.E., 1984, Diagenesis 7. Catagenesis of argillaceous sedimentary rocks: Geoscience Canada, v. 11, p. 67-75. Friedman, G.M., 1964, Early diagenesis and lithification in carbonate sediments: Journal of Sedimentary Petrology, v. 34, p. 777-813. Gatewood, L.E., 1970, Oklahoma City field - anatomy of a giant, in M.T. Halbouty, ed., Geology of Giant Petroleum Fields: AAPG Memoir 14, p. 223-254. Hall, J.F., 1990, The development of paleokarst and other solution features in the Mississippian Leadville Dolomite, central Colorado, in D.W. Beaty, G.P. Landis, and T.B. Thompson, eds., Carbonate-Hosted Sulfide Deposits of the Central Colorado Mineral Belt: Economic Geology Monograph 7, New Haven, Conn., Economic Geology Publ., p. 108-117. Halley, R.B., and Schmoker, J.W., 1983, High porosity Cenozoic carbonate rocks of south Florida: progressive loss of porosity with depth: AAPG Bulletin, v. 67, p. 191-200. Hanor, .S., 1987, Origin and migration of subsurface sedimentary brines: SEPM Lecture Notes, Short Course No. 21, 247 p. Hendrickson, A.R., Thomas, R.L., and Economides, M.J., 1992, Stimulation of carbonate reservoirs, in G.V. Chilingarian, S.J. Mazzullo, and H.H. Rieke, eds., Carbonate Reservoir Characterization: A Geologic-Engineering Analysis, Part I: Elsevier Publ. Co., Amsterdam, Developments in Petroleum Science 30, p. 589-625 . Heydari, E., and Moore, C.H., 1989, Burial diagenesis and thermochemical sulfate reduction, Smackover Formation, southeastern Mississippi salt basin: Geology, v. 17, p. 1080-1084. Hill, C.A., 1992, Sulfuric acid oil-field karst, in M.P. Candelaria and C.L. Reed, eds., Paleokarst, Karst Related Diagenesis and Reservoir Development: Examples from Ordovician-Devonian age strata of West Texas and the Mid-Continent: Permian Basin Section SEPM Publ. 92-33, p. 192-194. Holtz, M.H., and Kerans, C., 1992, Characterization and categorization of west Texas Ellenburger reservoirs, in M.P. Candelaria and C.L. Reed, eds., Paleokarst, Karst Related Diagenesis and Reservoir Development: Examples from Ordovician-Devonian age strata of West Texas and the Mid-Continent: Permian Basin Section SEPM Publ. 92-33, p. 45-54. Honarpour, M.M., Chilingarian, G.V., and Mazzullo, S.J., 1992, Permeability and relative permeability of carbonate reservoirs, in G.V. Chilingarian, S.J. Mazzullo, and H.H. Rieke, eds., Carbonate Reservoir Characterization: A Geologic-Engineering Analysis, Part I: Elsevier Publ. Co., Amsterdam, Developments in Petroleum Science 30, p. 399-416. Jodry, R.L., 1992, Pore geometry of carbonate rocks and capillary pressure curves (basic geologic concepts), in G.V. Chilingarian, S.J. Mazzullo, and H.H. Rieke, eds., Carbonate Reservoir Characterization: A Geologic-Engineering Analysis, Part I: Elsevier Publ. Co., Amsterdam, Developments in Petroleum Science 30, p. 331-377. Kharaka, Y.K., Law, L.M., Carothers, W.C., and Goerlitz, D.F., 1986, Role of organic species dissolved in formation waters from sedimentary basins in mineral diagenesis, in D.L. Gautier, ed., Roles of Organic Matter in Sediment Diagenesis: SEPM Special Publ. 38, p. 111-122. Land, L.S., 1967, Diagenesis of skeletal carbonates: Journal of Sedimentary Petrology, v. 37, p. 914-930. Longman, M.W., 1980, Carbonate diagenetic textures from near surface diagenetic environments: AAPG Bulletin, v. 64, p. 461-487. Lundegard, P.D., and Land, L.S., 1986, Carbon dioxide and inorganic acids: their role in porosity enhancement and cementation, Paloegene of the of the Texas Gulf Coast, in D.L. Gautier ed., Roles of Organic Matter in Sediment Diagenesis: SEPM Special Publ. 38, p. 129-146. Mazzullo, S.J., 1994, Models of porosity evolution in Permian periplatform carbonate reservoirs (debris-flows and turbidites) in the Permian Basin: West Texas Geological Society Bulletin, v. 34, no. 1, p. 5-12. Mazzullo, S.J., and Chilingarian, G.V., 1992, Diagenesis and origin of porosity, in G.V. Chilingarian, S.J. Mazzullo, and H.H. Rieke, eds., Carbonate Reservoir Characterization: A Geologic-Engineering Analysis, Part I: Elsevier Publ. Co., Amsterdam, Developments in Petroleum Science 30, p. 199-270.
Mazzullo, S.J., and Chilingarian, G.V.,
1996, Mazzullo, S.J., and Harris, P.M., 1991, An overview of dissolution porosity development in the deep-burial environment, with examples from carbonate reservoirs in the Permian Basin, in M. Candelaria, ed., Permian Basin Plays – Tomorrow’s Technology Today: West Texas Geological Society, Publ. No. 91-89, p. 125-138. Mazzullo, S.J., and Harris, P.M., 1992, Mesogenetic dissolution: its role in porosity development in carbonate reservoirs: AAPG Bulletin, v. 76, p. 607-620. Meshri, I.D., 1986, On the reactivity of carbonic and organic acids and generation of secondary porosity, in D. L. Gautier, ed., Roles of Organic Matter in Sediment Diagenesis: SEPM Special Publ. 38, p. 123-128. Moore, C.H., 1989, Carbonate Diagenesis and Porosity: Elsevier Publ. Co., Developments in Sedimentology 46, 338 p. Moore, C.H., and Druckman, Y., 1981, Burial diagenesis and porosity evolution, Upper Jurassic Smackover, Arkansas and Louisiana: AAPG Bulletin, v. 65, p. 597-628. Murray, R.C., 1960, Origin of porosity in carbonate rocks: Journal of Sedimentary Petrology, v. 30, p. 59-84. Newell, K.D., Watney, W.L., Cheng, S.W.L., and Brownrigg, R.L., 1987, Stratigraphic and spatial distribution of oil and gas production in Kansas: Kansas Geological Survey, Subsurface Geology Series 9, 86 p. Pittman, E.D., 1971, Microporosity in carbonate rocks: AAPG Bulletin, v. 55, p. 1873-1878. Radke, B.M., and Mathis, R.C., 1980, On the formation and occurrence of saddle dolomite: Journal of Sedimentary Petrology, v. 50, p. 1149-1168. Reeckmann, A., and Friedman, G.M., 1982, Exploration for Carbonate Petroleum Reservoirs: John Wiley and Sons, New York, 213 p. Roehl, P.O., 1985, Depositional and diagenetic controls on reservoir rock development and petrophysics in Silurian tidalites, Interlake Formation, Cabin Creek field area, Montana, in P.O. Roehl and P.W. Choquette, eds., Carbonate Petroleum Reservoirs: Springer-Verlag, Berlin, p. 85-105. Ruzyla, K., and Friedman, G.M., 1985, Factors controlling porosity in dolomite reservoirs of the Ordovician Red River Formation, Cabin Creek Field, Montana, in P.O. Roehl and P.W. Choquette, eds., Carbonate Petroleum Reservoirs: Springer-Verlag, Berlin, p. 41-58.
Sassen, R., and Moore, C.H., 1988,
Framework of Scholle, P.A., and Halley, R.B., 1985, Burial diagenesis: Out of sight, out of mind!, in N. Schneidermann and P. M. Harris, eds., Carbonate Cements: SEPM Special Publ. 36, p. 309-334. Surdam, R.C., Boese, S.W., and Crossey, L.J., 1984, The chemistry of secondary porosity, in D.A. McDonald and R.C. Surdam, eds., Clastic Diagenesis: AAPG Memoir 37, p. 127-149. Walters, R.F., 1946, Buried pre-Cambrian hills in northeastern Barton County, central Kansas: AAPG Bulletin, v. 30, p. 660-710.
Wardlaw, N.C., 1996, Factors affecting
oil Wardlaw, N.C., 1976, Pore geometry of carbonate rocks as revealed by pore casts and capillary pressures: AAPG Bulletin, v. 60, p. 245-257. Weyl, P.K., 1960, Porosity through dolomitization: conservation-of-mass requirements: Journal of Sedimentary Petrology, v. 30, p. 85-90. Wilhite, B.W., and Mazzullo, S.J., 2000, Facies architecture and diagenesis of Holocene carbonate sands in an inner-platform environment: analog of some ancient carbonate reservoirs, in S.T. Reid, ed., Transactions of Southwest Section AAPG, Publ. SWS 2000-107, p. 67-79. Wilhite, B.W., and Mazzullo, S.J., 2000, Facies architecture and diagenesis of Holocene carbonate sands in an inner-platform environment: analog of some ancient carbonate reservoirs, in S.T. Reid, ed., Transactions of Southwest Section AAPG, Publ. SWS 2000-107, p. 67-79. |